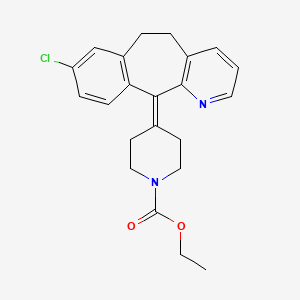
Loratadin
Übersicht
Beschreibung
Loratadine is a second-generation antihistamine widely used to manage symptoms of allergic rhinitis and urticaria (hives). It is known for its effectiveness in treating allergy symptoms such as sneezing, itching, and watery eyes without causing significant sedation, making it preferable over first-generation antihistamines .
Wirkmechanismus
Target of Action
Loratadine primarily targets the H1 histamine receptors . These receptors are found on the surface of various cells, including epithelial cells, endothelial cells, eosinophils, neutrophils, airway cells, and vascular smooth muscle cells . They play a crucial role in mediating allergic reactions.
Mode of Action
Loratadine acts as a selective inverse agonist for peripheral H1 histamine receptors . When an allergic reaction occurs, histamine is released and binds to these receptors, causing symptoms such as itching, sneezing, and runny nose. Loratadine intervenes by blocking this binding, effectively halting the allergic reaction .
Biochemical Pathways
Loratadine and its major metabolite, desloratadine, can extensively metabolize to hydroxylated metabolites (6-OH-DL, 5-OH-DL, and 3-OH-DL) by decarboethoxylation and subsequent oxidation and conjugation with glucuronic acid in vivo . These metabolites are also active and have a tendency for distributing to specific immune-regulatory tissues .
Pharmacokinetics
After ingestion, loratadine is rapidly absorbed, with its effects kicking in within 1-2 hours . It is then metabolized to its active form, desloratadine, which contributes significantly to allergy relief . Loratadine is metabolized by cytochrome P450 3A4 (CYP3A4) and, to a lesser extent, by cytochrome P450 2D6 (CYP2D6) . The body’s exposure to active metabolites is much higher than to the prodrug with loratadine .
Result of Action
Loratadine has been found to influence a range of cellular pathways, notably those pertaining to the cell cycle, cell senescence, P53 signaling pathway, and apoptosis . It can concurrently induce cell senescence and apoptosis . At higher concentrations, loratadine can trigger pyroptosis . It also reduces the expression of pro-inflammatory genes, including MMP1, MMP3, and MMP9, and inhibits AP-1 transcriptional activation .
Action Environment
The efficacy of loratadine can be influenced by environmental factors. For instance, in an Environmental Exposure Unit (EEU) study, the onset of action of loratadine tablets was found to be 75 minutes for the relief of nasal and ocular symptoms in adults with seasonal allergic rhinitis . This suggests that the environment, such as the presence of allergens, can influence the action and efficacy of loratadine.
Wissenschaftliche Forschungsanwendungen
Loratadin hat eine breite Palette von Anwendungen in der wissenschaftlichen Forschung:
5. Wirkmechanismus
This compound wirkt als selektiver inverser Agonist für periphere Histamin-H1-Rezeptoren. Wenn Histamin während einer allergischen Reaktion freigesetzt wird, bindet es an diese Rezeptoren und verursacht Symptome wie Juckreiz und Niesen. This compound blockiert diese Bindung und stoppt so effektiv die allergische Reaktion . Es hat minimale Auswirkungen auf das zentrale Nervensystem, wodurch das Risiko einer Sedierung verringert wird .
Biochemische Analyse
Biochemical Properties
Loratadine functions primarily by binding to histamine H1 receptors, which are G-protein coupled receptors located on the surface of various cells, including epithelial cells, endothelial cells, eosinophils, neutrophils, airway cells, and vascular smooth muscle cells . By binding to these receptors, loratadine prevents histamine from exerting its effects, thereby reducing allergic symptoms. The interaction between loratadine and the H1 receptor is characterized by its high affinity and selectivity, which contributes to its effectiveness in blocking histamine-induced responses .
Cellular Effects
Loratadine exerts several effects on different cell types and cellular processes. It has been shown to influence cell signaling pathways, gene expression, and cellular metabolism. For instance, loratadine can modulate the activity of various signaling molecules involved in inflammatory responses, such as cytokines and chemokines . Additionally, loratadine has been associated with improved prognosis in certain cancers, such as lung cancer, by inducing apoptosis and reducing epithelial-mesenchymal transition . These effects are mediated through its interaction with specific cellular receptors and signaling pathways, highlighting its potential therapeutic benefits beyond allergy management.
Molecular Mechanism
At the molecular level, loratadine exerts its effects by acting as an inverse agonist of the histamine H1 receptor . This means that loratadine not only blocks the binding of histamine to the receptor but also stabilizes the receptor in its inactive state, thereby reducing its basal activity. The binding of loratadine to the H1 receptor involves interactions with specific amino acid residues within the receptor’s binding pocket, which prevents the conformational changes required for receptor activation . This mechanism of action underlies the antihistaminic and anti-inflammatory effects of loratadine.
Temporal Effects in Laboratory Settings
In laboratory settings, the effects of loratadine have been studied over various time periods to assess its stability, degradation, and long-term impact on cellular function. Loratadine is known to have a relatively long half-life, which contributes to its sustained therapeutic effects . Studies have shown that loratadine and its active metabolite, desloratadine, can maintain their efficacy over extended periods, with minimal degradation . Additionally, long-term exposure to loratadine has been associated with consistent anti-inflammatory and antihistaminic effects, further supporting its use in chronic allergic conditions .
Dosage Effects in Animal Models
The effects of loratadine at different dosages have been extensively studied in animal models. These studies have revealed that loratadine exhibits dose-dependent effects, with higher doses leading to more pronounced therapeutic outcomes . Excessive dosages can result in adverse effects, such as drowsiness, dry mouth, and gastrointestinal disturbances . In cancer models, moderate concentrations of loratadine have been shown to induce apoptosis and reduce epithelial-mesenchymal transition, while higher concentrations can trigger pyroptosis . These findings highlight the importance of optimizing loratadine dosage to achieve the desired therapeutic effects while minimizing potential side effects.
Metabolic Pathways
Loratadine undergoes extensive first-pass metabolism in the liver, primarily mediated by cytochrome P450 enzymes, including CYP3A4, CYP2D6, CYP1A1, and CYP2C19 . The major metabolite of loratadine is desloratadine, which retains antihistaminic activity and contributes to the overall therapeutic effects of the drug . The metabolic pathways involved in loratadine metabolism also include hydroxylation and conjugation reactions, which facilitate the elimination of the drug from the body . Understanding these metabolic pathways is crucial for optimizing loratadine dosing and minimizing potential drug interactions.
Transport and Distribution
Loratadine is transported and distributed within cells and tissues through various mechanisms. It binds to plasma proteins, which facilitates its distribution throughout the body . The tissue distribution of loratadine and its metabolites has been studied in animal models, revealing that they are widely distributed in organs such as the liver, spleen, thymus, heart, adrenal glands, and pituitary gland . The concentrations of loratadine and its metabolites in these tissues are influenced by factors such as blood flow, tissue permeability, and binding affinity to cellular receptors . These findings provide insights into the pharmacokinetics and tissue-specific effects of loratadine.
Subcellular Localization
The subcellular localization of loratadine and its effects on cellular activity have been investigated to understand its precise mechanism of action. Loratadine is known to localize to specific cellular compartments, such as the plasma membrane and cytoplasm, where it interacts with histamine H1 receptors . The targeting of loratadine to these compartments is facilitated by its chemical structure and binding properties, which enable it to effectively block histamine-induced signaling pathways . Additionally, loratadine may undergo post-translational modifications that influence its localization and activity within cells
Vorbereitungsmethoden
Synthetic Routes and Reaction Conditions: Loratadine can be synthesized through various methods. One common method involves the reaction of ethyl 4-(8-chloro-5,6-dihydro-11H-benzo[5,6]cyclohepta[1,2-b]pyridin-11-ylidene)-1-piperidinecarboxylate with appropriate reagents under controlled conditions. The process typically includes steps such as dissolving crude loratadine in an organic solvent, adding active carbon for adsorption, heating, stirring, and filtering to obtain purified loratadine .
Industrial Production Methods: In industrial settings, loratadine is produced using high-speed shear-high pressure homogenization followed by freeze-drying to create loratadine nanocrystals. This method enhances the solubility and bioavailability of loratadine, making it more effective for oral administration .
Analyse Chemischer Reaktionen
Reaktionstypen: Loratadin unterliegt verschiedenen chemischen Reaktionen, darunter Oxidation, Reduktion und Substitution. Beispielsweise ist eine Oxidation in den Piperidin- und Cycloheptanringen wahrscheinlich .
Häufige Reagenzien und Bedingungen: Zu den häufig verwendeten Reagenzien in diesen Reaktionen gehören Wasserstoffperoxid zur Oxidation und Natriumborhydrid zur Reduktion. Die Bedingungen umfassen typischerweise kontrollierte Temperaturen und pH-Werte, um die gewünschten Reaktionsergebnisse sicherzustellen.
Geformte Hauptprodukte: Zu den Hauptprodukten, die aus diesen Reaktionen entstehen, gehört Desthis compound, ein aktiver Metabolit von this compound, der antihistaminische Eigenschaften behält .
Vergleich Mit ähnlichen Verbindungen
Loratadin wird häufig mit anderen Antihistaminika der zweiten Generation wie Cetirizin und Fexofenadin verglichen. Im Gegensatz zu Antihistaminika der ersten Generation wie Diphenhydramin überwindet this compound die Blut-Hirn-Schranke nicht signifikant, was zu weniger sedierenden Wirkungen führt . Ähnliche Verbindungen sind:
Fexofenadin: Ein weiteres nicht sedierendes Antihistaminikum mit einem ähnlichen Wirkmechanismus.
Chlorpheniramin: Ein Antihistaminikum der ersten Generation, das wirksam ist, aber eine signifikante Sedierung verursacht.
Der einzigartige Vorteil von this compound liegt in seiner Fähigkeit, eine wirksame Linderung von Allergien zu bieten, ohne Schläfrigkeit zu verursachen, was es für viele Patienten zur bevorzugten Wahl macht.
Eigenschaften
IUPAC Name |
ethyl 4-(13-chloro-4-azatricyclo[9.4.0.03,8]pentadeca-1(11),3(8),4,6,12,14-hexaen-2-ylidene)piperidine-1-carboxylate | |
|---|---|---|
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
InChI |
InChI=1S/C22H23ClN2O2/c1-2-27-22(26)25-12-9-15(10-13-25)20-19-8-7-18(23)14-17(19)6-5-16-4-3-11-24-21(16)20/h3-4,7-8,11,14H,2,5-6,9-10,12-13H2,1H3 | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
InChI Key |
JCCNYMKQOSZNPW-UHFFFAOYSA-N | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
Canonical SMILES |
CCOC(=O)N1CCC(=C2C3=C(CCC4=C2N=CC=C4)C=C(C=C3)Cl)CC1 | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
Molecular Formula |
C22H23ClN2O2 | |
| Record name | loratadine | |
| Source | Wikipedia | |
| URL | https://en.wikipedia.org/wiki/Loratadine | |
| Description | Chemical information link to Wikipedia. | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
DSSTOX Substance ID |
DTXSID2023224 | |
| Record name | Loratadine | |
| Source | EPA DSSTox | |
| URL | https://comptox.epa.gov/dashboard/DTXSID2023224 | |
| Description | DSSTox provides a high quality public chemistry resource for supporting improved predictive toxicology. | |
Molecular Weight |
382.9 g/mol | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
Physical Description |
Solid | |
| Record name | Loratadine | |
| Source | Human Metabolome Database (HMDB) | |
| URL | http://www.hmdb.ca/metabolites/HMDB0005000 | |
| Description | The Human Metabolome Database (HMDB) is a freely available electronic database containing detailed information about small molecule metabolites found in the human body. | |
| Explanation | HMDB is offered to the public as a freely available resource. Use and re-distribution of the data, in whole or in part, for commercial purposes requires explicit permission of the authors and explicit acknowledgment of the source material (HMDB) and the original publication (see the HMDB citing page). We ask that users who download significant portions of the database cite the HMDB paper in any resulting publications. | |
Solubility |
<1 mg/ml at 25°C | |
| Record name | Loratadine | |
| Source | DrugBank | |
| URL | https://www.drugbank.ca/drugs/DB00455 | |
| Description | The DrugBank database is a unique bioinformatics and cheminformatics resource that combines detailed drug (i.e. chemical, pharmacological and pharmaceutical) data with comprehensive drug target (i.e. sequence, structure, and pathway) information. | |
| Explanation | Creative Common's Attribution-NonCommercial 4.0 International License (http://creativecommons.org/licenses/by-nc/4.0/legalcode) | |
Mechanism of Action |
Histamine release is a key mediator in allergic rhinitis and urticaria. As a result, loratadine exerts it's effect by targeting H1 histamine receptors. Loratadine binds to H1 histamine receptors found on the surface of epithelial cells, endothelial cells, eosinophils, neutrophils, airway cells, and vascular smooth muscle cells among others. H1 histamine receptors fall under the wider umbrella of G-protein coupled receptors, and exist in a state of equilibrium between the active and inactive forms. Histamine binding to the H1-receptor facilitates cross linking between transmembrane domains III and V, stabilizing the active form of the receptor. On the other hand, antihistamines bind to a different site on the H1 receptor favouring the inactive form. Hence, loratadine can more accurately be classified as an "inverse agonist" as opposed to a "histamine antagonist", and can prevent or reduce the severity of histamine mediated symptoms., All of the available H1 receptor antagonists are reversible, competitive inhibitors of the interaction of histamine with H1 receptors. /H1 Receptor Antagonists/, H1 antagonists inhibit most responses of smooth muscle to histamine. /H1 Antagonists Receptors/, Within the vascular tree, the H1 antagonists inhibit both the vasoconstrictor effects of histamine and, to a degree, the more rapid vasodilator effects that are mediated by H1 receptors on endothelial cells. /H1 Receptor Antagonists/, H1 antagonists strongly block the action of histamine that results in increased capillary permeability and formation of edema and wheal. /H1 Receptor Antagonists/, For more Mechanism of Action (Complete) data for LORATADINE (6 total), please visit the HSDB record page. | |
| Record name | Loratadine | |
| Source | DrugBank | |
| URL | https://www.drugbank.ca/drugs/DB00455 | |
| Description | The DrugBank database is a unique bioinformatics and cheminformatics resource that combines detailed drug (i.e. chemical, pharmacological and pharmaceutical) data with comprehensive drug target (i.e. sequence, structure, and pathway) information. | |
| Explanation | Creative Common's Attribution-NonCommercial 4.0 International License (http://creativecommons.org/licenses/by-nc/4.0/legalcode) | |
| Record name | LORATADINE | |
| Source | Hazardous Substances Data Bank (HSDB) | |
| URL | https://pubchem.ncbi.nlm.nih.gov/source/hsdb/3578 | |
| Description | The Hazardous Substances Data Bank (HSDB) is a toxicology database that focuses on the toxicology of potentially hazardous chemicals. It provides information on human exposure, industrial hygiene, emergency handling procedures, environmental fate, regulatory requirements, nanomaterials, and related areas. The information in HSDB has been assessed by a Scientific Review Panel. | |
Color/Form |
Crystals from acetonitrile | |
CAS No. |
79794-75-5 | |
| Record name | Loratadine | |
| Source | CAS Common Chemistry | |
| URL | https://commonchemistry.cas.org/detail?cas_rn=79794-75-5 | |
| Description | CAS Common Chemistry is an open community resource for accessing chemical information. Nearly 500,000 chemical substances from CAS REGISTRY cover areas of community interest, including common and frequently regulated chemicals, and those relevant to high school and undergraduate chemistry classes. This chemical information, curated by our expert scientists, is provided in alignment with our mission as a division of the American Chemical Society. | |
| Explanation | The data from CAS Common Chemistry is provided under a CC-BY-NC 4.0 license, unless otherwise stated. | |
| Record name | Loratadine [USAN:USP:INN:BAN] | |
| Source | ChemIDplus | |
| URL | https://pubchem.ncbi.nlm.nih.gov/substance/?source=chemidplus&sourceid=0079794755 | |
| Description | ChemIDplus is a free, web search system that provides access to the structure and nomenclature authority files used for the identification of chemical substances cited in National Library of Medicine (NLM) databases, including the TOXNET system. | |
| Record name | Loratadine | |
| Source | DrugBank | |
| URL | https://www.drugbank.ca/drugs/DB00455 | |
| Description | The DrugBank database is a unique bioinformatics and cheminformatics resource that combines detailed drug (i.e. chemical, pharmacological and pharmaceutical) data with comprehensive drug target (i.e. sequence, structure, and pathway) information. | |
| Explanation | Creative Common's Attribution-NonCommercial 4.0 International License (http://creativecommons.org/licenses/by-nc/4.0/legalcode) | |
| Record name | loratadine | |
| Source | DTP/NCI | |
| URL | https://dtp.cancer.gov/dtpstandard/servlet/dwindex?searchtype=NSC&outputformat=html&searchlist=758628 | |
| Description | The NCI Development Therapeutics Program (DTP) provides services and resources to the academic and private-sector research communities worldwide to facilitate the discovery and development of new cancer therapeutic agents. | |
| Explanation | Unless otherwise indicated, all text within NCI products is free of copyright and may be reused without our permission. Credit the National Cancer Institute as the source. | |
| Record name | loratadine | |
| Source | DTP/NCI | |
| URL | https://dtp.cancer.gov/dtpstandard/servlet/dwindex?searchtype=NSC&outputformat=html&searchlist=721075 | |
| Description | The NCI Development Therapeutics Program (DTP) provides services and resources to the academic and private-sector research communities worldwide to facilitate the discovery and development of new cancer therapeutic agents. | |
| Explanation | Unless otherwise indicated, all text within NCI products is free of copyright and may be reused without our permission. Credit the National Cancer Institute as the source. | |
| Record name | Loratadine | |
| Source | EPA DSSTox | |
| URL | https://comptox.epa.gov/dashboard/DTXSID2023224 | |
| Description | DSSTox provides a high quality public chemistry resource for supporting improved predictive toxicology. | |
| Record name | ethyl 4-(8-chloro-5,6-dihydro-11H-benzo[5,6]cyclohepta[1,2-b]pyridin-11-ylidene)piperidine-1-carboxylate | |
| Source | European Chemicals Agency (ECHA) | |
| URL | https://echa.europa.eu/substance-information/-/substanceinfo/100.216.235 | |
| Description | The European Chemicals Agency (ECHA) is an agency of the European Union which is the driving force among regulatory authorities in implementing the EU's groundbreaking chemicals legislation for the benefit of human health and the environment as well as for innovation and competitiveness. | |
| Explanation | Use of the information, documents and data from the ECHA website is subject to the terms and conditions of this Legal Notice, and subject to other binding limitations provided for under applicable law, the information, documents and data made available on the ECHA website may be reproduced, distributed and/or used, totally or in part, for non-commercial purposes provided that ECHA is acknowledged as the source: "Source: European Chemicals Agency, http://echa.europa.eu/". Such acknowledgement must be included in each copy of the material. ECHA permits and encourages organisations and individuals to create links to the ECHA website under the following cumulative conditions: Links can only be made to webpages that provide a link to the Legal Notice page. | |
| Record name | 1-Piperidinecarboxylic acid, 4-(8-chloro-5,6-dihydro-11H-benzo[5,6]cyclohepta[1,2-b]pyridin-11-ylidene)-, ethyl ester | |
| Source | European Chemicals Agency (ECHA) | |
| URL | https://echa.europa.eu/substance-information/-/substanceinfo/100.120.122 | |
| Description | The European Chemicals Agency (ECHA) is an agency of the European Union which is the driving force among regulatory authorities in implementing the EU's groundbreaking chemicals legislation for the benefit of human health and the environment as well as for innovation and competitiveness. | |
| Explanation | Use of the information, documents and data from the ECHA website is subject to the terms and conditions of this Legal Notice, and subject to other binding limitations provided for under applicable law, the information, documents and data made available on the ECHA website may be reproduced, distributed and/or used, totally or in part, for non-commercial purposes provided that ECHA is acknowledged as the source: "Source: European Chemicals Agency, http://echa.europa.eu/". Such acknowledgement must be included in each copy of the material. ECHA permits and encourages organisations and individuals to create links to the ECHA website under the following cumulative conditions: Links can only be made to webpages that provide a link to the Legal Notice page. | |
| Record name | LORATADINE | |
| Source | FDA Global Substance Registration System (GSRS) | |
| URL | https://gsrs.ncats.nih.gov/ginas/app/beta/substances/7AJO3BO7QN | |
| Description | The FDA Global Substance Registration System (GSRS) enables the efficient and accurate exchange of information on what substances are in regulated products. Instead of relying on names, which vary across regulatory domains, countries, and regions, the GSRS knowledge base makes it possible for substances to be defined by standardized, scientific descriptions. | |
| Explanation | Unless otherwise noted, the contents of the FDA website (www.fda.gov), both text and graphics, are not copyrighted. They are in the public domain and may be republished, reprinted and otherwise used freely by anyone without the need to obtain permission from FDA. Credit to the U.S. Food and Drug Administration as the source is appreciated but not required. | |
| Record name | LORATADINE | |
| Source | Hazardous Substances Data Bank (HSDB) | |
| URL | https://pubchem.ncbi.nlm.nih.gov/source/hsdb/3578 | |
| Description | The Hazardous Substances Data Bank (HSDB) is a toxicology database that focuses on the toxicology of potentially hazardous chemicals. It provides information on human exposure, industrial hygiene, emergency handling procedures, environmental fate, regulatory requirements, nanomaterials, and related areas. The information in HSDB has been assessed by a Scientific Review Panel. | |
| Record name | Loratadine | |
| Source | Human Metabolome Database (HMDB) | |
| URL | http://www.hmdb.ca/metabolites/HMDB0005000 | |
| Description | The Human Metabolome Database (HMDB) is a freely available electronic database containing detailed information about small molecule metabolites found in the human body. | |
| Explanation | HMDB is offered to the public as a freely available resource. Use and re-distribution of the data, in whole or in part, for commercial purposes requires explicit permission of the authors and explicit acknowledgment of the source material (HMDB) and the original publication (see the HMDB citing page). We ask that users who download significant portions of the database cite the HMDB paper in any resulting publications. | |
Melting Point |
134-136 °C, 134 - 136 °C | |
| Record name | Loratadine | |
| Source | DrugBank | |
| URL | https://www.drugbank.ca/drugs/DB00455 | |
| Description | The DrugBank database is a unique bioinformatics and cheminformatics resource that combines detailed drug (i.e. chemical, pharmacological and pharmaceutical) data with comprehensive drug target (i.e. sequence, structure, and pathway) information. | |
| Explanation | Creative Common's Attribution-NonCommercial 4.0 International License (http://creativecommons.org/licenses/by-nc/4.0/legalcode) | |
| Record name | LORATADINE | |
| Source | Hazardous Substances Data Bank (HSDB) | |
| URL | https://pubchem.ncbi.nlm.nih.gov/source/hsdb/3578 | |
| Description | The Hazardous Substances Data Bank (HSDB) is a toxicology database that focuses on the toxicology of potentially hazardous chemicals. It provides information on human exposure, industrial hygiene, emergency handling procedures, environmental fate, regulatory requirements, nanomaterials, and related areas. The information in HSDB has been assessed by a Scientific Review Panel. | |
| Record name | Loratadine | |
| Source | Human Metabolome Database (HMDB) | |
| URL | http://www.hmdb.ca/metabolites/HMDB0005000 | |
| Description | The Human Metabolome Database (HMDB) is a freely available electronic database containing detailed information about small molecule metabolites found in the human body. | |
| Explanation | HMDB is offered to the public as a freely available resource. Use and re-distribution of the data, in whole or in part, for commercial purposes requires explicit permission of the authors and explicit acknowledgment of the source material (HMDB) and the original publication (see the HMDB citing page). We ask that users who download significant portions of the database cite the HMDB paper in any resulting publications. | |
Synthesis routes and methods I
Procedure details









Synthesis routes and methods II
Procedure details








Retrosynthesis Analysis
AI-Powered Synthesis Planning: Our tool employs the Template_relevance Pistachio, Template_relevance Bkms_metabolic, Template_relevance Pistachio_ringbreaker, Template_relevance Reaxys, Template_relevance Reaxys_biocatalysis model, leveraging a vast database of chemical reactions to predict feasible synthetic routes.
One-Step Synthesis Focus: Specifically designed for one-step synthesis, it provides concise and direct routes for your target compounds, streamlining the synthesis process.
Accurate Predictions: Utilizing the extensive PISTACHIO, BKMS_METABOLIC, PISTACHIO_RINGBREAKER, REAXYS, REAXYS_BIOCATALYSIS database, our tool offers high-accuracy predictions, reflecting the latest in chemical research and data.
Strategy Settings
| Precursor scoring | Relevance Heuristic |
|---|---|
| Min. plausibility | 0.01 |
| Model | Template_relevance |
| Template Set | Pistachio/Bkms_metabolic/Pistachio_ringbreaker/Reaxys/Reaxys_biocatalysis |
| Top-N result to add to graph | 6 |
Feasible Synthetic Routes
Haftungsausschluss und Informationen zu In-Vitro-Forschungsprodukten
Bitte beachten Sie, dass alle Artikel und Produktinformationen, die auf BenchChem präsentiert werden, ausschließlich zu Informationszwecken bestimmt sind. Die auf BenchChem zum Kauf angebotenen Produkte sind speziell für In-vitro-Studien konzipiert, die außerhalb lebender Organismen durchgeführt werden. In-vitro-Studien, abgeleitet von dem lateinischen Begriff "in Glas", beinhalten Experimente, die in kontrollierten Laborumgebungen unter Verwendung von Zellen oder Geweben durchgeführt werden. Es ist wichtig zu beachten, dass diese Produkte nicht als Arzneimittel oder Medikamente eingestuft sind und keine Zulassung der FDA für die Vorbeugung, Behandlung oder Heilung von medizinischen Zuständen, Beschwerden oder Krankheiten erhalten haben. Wir müssen betonen, dass jede Form der körperlichen Einführung dieser Produkte in Menschen oder Tiere gesetzlich strikt untersagt ist. Es ist unerlässlich, sich an diese Richtlinien zu halten, um die Einhaltung rechtlicher und ethischer Standards in Forschung und Experiment zu gewährleisten.
![N-[(3S,4R,5S,9R,10R,13R,14R,17R)-4-hydroxy-10,13-dimethyl-17-[(2R)-1-(4-propan-2-ylidene-2,3-dihydropyrrol-5-yl)propan-2-yl]-2,3,4,5,6,9,11,12,14,15,16,17-dodecahydro-1H-cyclopenta[a]phenanthren-3-yl]acetamide](/img/structure/B1675032.png)
![(1S,9R,13R,19S,22S,23S,25R,26R,28S,31S,33S,36R)-22-Hydroxy-1,10,10,12,12,30,30,36-octamethyl-28-(2-methylprop-1-enyl)-11,24,27,29,32-pentaoxa-3-azadecacyclo[17.17.0.02,17.04,16.07,15.09,13.022,36.023,25.023,33.026,31]hexatriaconta-2(17),4(16),5,7(15)-tetraen-8-one](/img/structure/B1675035.png)
![2-[(3R,6S,9S,12S,15S,18S,21S,24R,27S,30S)-21-(2-amino-2-oxoethyl)-9-(3-aminopropyl)-24,27-dibenzyl-3-[(4-hydroxyphenyl)methyl]-15-(1H-indol-3-ylmethyl)-6-(2-methylpropyl)-2,5,8,11,14,17,20,23,26,29-decaoxo-12-propyl-1,4,7,10,13,16,19,22,25,28-decazabicyclo[28.3.0]tritriacontan-18-yl]acetic acid](/img/structure/B1675036.png)
