Sulfate de baryum
Vue d'ensemble
Description
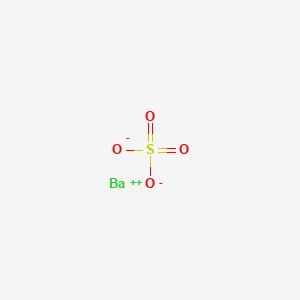
Barium sulfate is an inorganic compound with the chemical formula BaSO₄. It is a white crystalline solid that is odorless and insoluble in water. This compound occurs naturally as the mineral barite, which is the primary commercial source of barium and its derivatives . Barium sulfate is known for its high density and opacity, making it useful in various industrial and medical applications .
Synthetic Routes and Reaction Conditions:
-
Using Barium Chloride and Sulfuric Acid:
-
Using Barium Sulfide and Sodium Sulfate:
Industrial Production Methods:
Direct Precipitation: This method involves the interaction of barium ions and sulfate ions in a liquid environment.
Types of Reactions:
Precipitation Reaction: Barium sulfate is formed as a precipitate when barium ions react with sulfate ions in an aqueous solution.
Common Reagents and Conditions:
Sulfuric Acid and Barium Chloride: Used in the precipitation reaction to form barium sulfate.
Sodium Sulfate and Barium Sulfide: Alternative reagents for the precipitation of barium sulfate.
Major Products:
Applications De Recherche Scientifique
Barium sulfate has a wide range of applications in scientific research:
Mécanisme D'action
Target of Action
Barium sulfate is primarily used as a radiographic contrast agent for X-ray imaging and other diagnostic procedures . Its primary targets are the structures within the gastrointestinal (GI) tract .
Mode of Action
Barium sulfate works by increasing the absorption of X-rays as they pass through the body . Due to its high atomic number, barium is opaque to X-rays, which allows for the clear visualization of structures where barium sulfate is localized . This compound is ingested or administered rectally and combined with granules of effervescent bicarbonate to enhance distension of the GI tract, allowing for enhanced visualization .
Biochemical Pathways
It remains in the GI tract without being absorbed into the bloodstream . It’s worth noting that barium ions can interfere with potassium channels in the body, affecting muscle and nerve function .
Pharmacokinetics
Barium sulfate exhibits unique pharmacokinetic properties due to its low water solubility and high level of clearance from the body . After administration, it is excreted from the body through the rectum .
Result of Action
The primary result of barium sulfate’s action is the clear visualization of the GI tract during X-ray imaging . It allows healthcare professionals to diagnose and monitor various conditions within the GI tract .
Action Environment
The action of barium sulfate can be influenced by environmental factors. For instance, the presence of other elements in the GI tract can affect the distribution and excretion of barium sulfate . Additionally, the compound’s action may be influenced by the patient’s hydration status, as adequate hydration can help avoid obstruction or impaction following a barium sulfate procedure .
Analyse Biochimique
Biochemical Properties
Barium Sulfate does not directly participate in biochemical reactions. It is primarily used in medical procedures and industrial applications due to its physical properties . It can enhance the fluorescence properties of organic dyes, making it a valuable tool in food safety and quality control .
Cellular Effects
Its primary use in the biological context is as a contrast agent in medical imaging, where it helps visualize certain structures or areas in the body .
Molecular Mechanism
The molecular mechanism of Barium Sulfate primarily involves its interaction with X-rays during medical imaging. As a heavy metal compound, Barium Sulfate absorbs X-rays and appears white on an X-ray film, helping to highlight the gastrointestinal tract .
Temporal Effects in Laboratory Settings
In laboratory settings, Barium Sulfate is stable and does not degrade over time . It does not have any long-term effects on cellular function observed in in vitro or in vivo studies .
Dosage Effects in Animal Models
The effects of Barium Sulfate in animal models are primarily related to its use as a contrast agent in medical imaging. The compound is generally considered safe at the dosages used for this purpose .
Metabolic Pathways
Barium Sulfate is not involved in any known metabolic pathways. It is not metabolized by the body and is excreted unchanged .
Transport and Distribution
After oral administration, Barium Sulfate is transported through the digestive tract. It is not absorbed into the body but is eliminated unchanged in the feces .
Subcellular Localization
Barium Sulfate does not enter cells and therefore does not have a subcellular localization. It remains in the gastrointestinal tract after administration and is excreted unchanged .
Comparaison Avec Des Composés Similaires
Calcium Sulfate (CaSO₄): Like barium sulfate, calcium sulfate is also used in medical imaging but has different sol
Propriétés
| Barium sulfate is a heavy metal with a high atomic number (Z=56) and a K shell binding energy (K-edge of 37.4 keV) very close to that of most diagnostic x-ray beams. Due to these characteristics, barium is an ideal medium for the absorption of x-rays. Barium sulfate is essentially not absorbed from the GI tract nor metabolized in the body. Barium sulfate is used to fill the gastrointestinal tract lumen or to coat the mucosal surface and is administered orally, rectally, or instilled into an enterostomy tube or catheter,. Barium sulfate enhances delineation of the GI tract. The barium suspension covers the mucosal surface of the GI tract, allowing its shape, distensibility, motion, integrity, continuity, location within the torso, relationship to other organs to be closely examined. Various abnormalities, such as benign or malignant tumors, ulcers, strictures, diverticula, inflammation or infection, altered motility, displacement and other pathology can thereby be identified,. At lower concentrations (higher dilution), barium enhances the conspicuity of the GI tract to differentiate the GI tract from various abdominal organs in computed tomography examinations (CT scans) of the abdomen. Improved delineation of the gastrointestinal tract lumen and mucosa may be reached by contrast provided by gas (by the addition of bicarbonate or gas-filled balloons) in addition to the barium. This is known as a _double-contrast procedure_. Osmotically active agents (for example, sorbitol) are also used to induce fluid accumulation and distension of the GI system to enhance visualization. | |
Numéro CAS |
7727-43-7 |
Formule moléculaire |
BaH2O4S |
Poids moléculaire |
235.41 g/mol |
Nom IUPAC |
barium(2+);sulfate |
InChI |
InChI=1S/Ba.H2O4S/c;1-5(2,3)4/h;(H2,1,2,3,4) |
Clé InChI |
XUBKPYAWPSXPDZ-UHFFFAOYSA-N |
Impuretés |
Natural impurities are ferric oxide, silicon dioxide, and strontium sulfate. |
SMILES |
[O-]S(=O)(=O)[O-].[Ba+2] |
SMILES canonique |
OS(=O)(=O)O.[Ba] |
Point d'ébullition |
2912 °F at 760 mmHg (Decomposes) (NIOSH, 2024) decomposes 2912 °F (decomposes) 2912 °F (Decomposes) |
Color/Form |
Fine, heavy powder or polymorphous crystals White, orthorhombic crystals White or yellowish powder Opaque powde |
Densité |
4.25 to 4.5 (NIOSH, 2024) 4.49 g/cu cm 4.5 g/cm³ 4.25-4.5 |
melting_point |
2876 °F (NIOSH, 2024) 1580 °C 2876 °F |
| 7727-43-7 13462-86-7 |
|
Description physique |
Barium sulfate appears as white or yellowish odorless powder or small crystals. Mp: 1580 °C (with decomposition). Density: 4.25 -4.5 g cm-3. Insoluble in water, dilute acids, alcohol. Soluble in hot concentrated sulfuric acid. Used as a weighting mud in oil-drilling, in paints, paper coatings, linoleum, textiles, rubber. Administered internally ("barium cocktail") as a radio-opaque diagnostic aid. Pellets or Large Crystals; Dry Powder; NKRA; Water or Solvent Wet Solid; Other Solid; Dry Powder, Liquid; Liquid, Other Solid; Liquid White or yellowish, odorless powder; [NIOSH] Insoluble in water; [HSDB] ODOURLESS TASTELESS WHITE OR YELLOWISH CRYSTALS OR POWDER. White or yellowish, odorless powder. |
Pictogrammes |
Health Hazard |
Solubilité |
0.0002 % at 64 °F (NIOSH, 2024) Very slightly soluble in cold water SOLUBILITY IN WATER INCREASES CONSIDERABLY IN PRESENCE OF CHLORIDE & OTHER ANIONS 0.00031 g/100 g water at 20 °C; insol in ethanol Soluble in hot concentrated sulfuric acid; practically insoluble in dilute acids and alcohol Practically insoluble in organic solvents; very slightly soluble in alkalis and in solution of many salts Solubility in water: none (64 °F): 0.0002% |
Synonymes |
Barite Baritop Barium Sulfate Barium Sulfate (2:1) E Z CAT E-Z-CAT EZCAT Micropaque Oral Sulfate, Barium |
Pression de vapeur |
0 mmHg (approx) (NIOSH, 2024) 0 mmHg (approx) |
Origine du produit |
United States |
Synthesis routes and methods I
Procedure details









Synthesis routes and methods II
Procedure details








Synthesis routes and methods III
Procedure details








Synthesis routes and methods IV
Procedure details










Retrosynthesis Analysis
AI-Powered Synthesis Planning: Our tool employs the Template_relevance Pistachio, Template_relevance Bkms_metabolic, Template_relevance Pistachio_ringbreaker, Template_relevance Reaxys, Template_relevance Reaxys_biocatalysis model, leveraging a vast database of chemical reactions to predict feasible synthetic routes.
One-Step Synthesis Focus: Specifically designed for one-step synthesis, it provides concise and direct routes for your target compounds, streamlining the synthesis process.
Accurate Predictions: Utilizing the extensive PISTACHIO, BKMS_METABOLIC, PISTACHIO_RINGBREAKER, REAXYS, REAXYS_BIOCATALYSIS database, our tool offers high-accuracy predictions, reflecting the latest in chemical research and data.
Strategy Settings
| Precursor scoring | Relevance Heuristic |
|---|---|
| Min. plausibility | 0.01 |
| Model | Template_relevance |
| Template Set | Pistachio/Bkms_metabolic/Pistachio_ringbreaker/Reaxys/Reaxys_biocatalysis |
| Top-N result to add to graph | 6 |
Feasible Synthetic Routes
Q1: What is the molecular formula and weight of barium sulfate?
A1: Barium sulfate has the molecular formula BaSO4 and a molecular weight of 233.38 g/mol.
Q2: What are some key properties of barium sulfate?
A2: Barium sulfate is an inorganic compound characterized by its high density, white color, and insolubility in water. It is chemically inert and thermally stable, decomposing at temperatures above 1600°C. These properties make it suitable for various applications.
Q3: How is barium sulfate used in medical imaging?
A3: Barium sulfate is widely used as a radiopaque contrast agent in medical imaging, particularly for visualizing the gastrointestinal tract. Due to its insolubility and high atomic number, it absorbs X-rays efficiently, allowing for clear visualization of the digestive system during procedures like barium swallows and barium enemas [, ].
Q4: Are there any concerns regarding the gluten content of barium sulfate suspensions used in medical procedures for patients with Celiac disease?
A4: Yes, determining the gluten content of commercially available barium sulfate suspensions can be challenging. A study revealed that a significant proportion of hospitals and imaging centers were unaware of the gluten content in the contrast media they used []. Clinicians need to ensure gluten-free barium sulfate suspensions are used for patients with Celiac disease or those following a gluten-free diet.
Q5: Can barium sulfate be used for imaging modalities other than X-ray?
A5: Research suggests barium sulfate can be combined with near-infrared-II (NIR-II) absorbing pigments to create a dual-modality contrast agent for both X-ray and photoacoustic imaging (PAI) []. This could allow for complementary imaging information from a single contrast administration.
Q6: Are there any risks associated with the use of barium sulfate in medical imaging?
A6: While generally considered safe, there have been isolated reports of complications related to barium sulfate use. In one instance, a hemodialysis catheter fracture was attributed to the accumulation of barium sulfate particles within the catheter material []. This emphasizes the importance of careful formulation and quality control in the manufacturing of medical-grade barium sulfate.
Q7: Can barium sulfate be used as a filler in polymer composites?
A7: Yes, barium sulfate can function as a filler in polymer composites. For example, incorporating barium sulfate into polypropylene/polystyrene blends can modify the blend's microstructure and dynamic mechanical properties []. The presence of specific modifiers, such as maleic anhydride-grafted polypropylene, can influence the filler's distribution within the polymer matrix, further affecting the composite's properties.
Q8: How does barium sulfate contribute to radiation shielding?
A8: Barium sulfate's high atomic number makes it effective for attenuating X-rays. Studies have explored its use in radiation-shielding materials. One study investigated incorporating barium sulfate into bricks, showing promising X-ray shielding properties with increasing barium sulfate content []. Another study developed X-ray-shielding films using barium sulfate nanoparticles embedded in a polyethylene terephthalate (PET) matrix, demonstrating potential as an alternative to lead-based shielding materials [].
Q9: Can barium sulfate be used to create colored pigments?
A9: Yes, barium sulfate can be used as a base material for producing pigments. One study describes a method of depositing ferric oxide onto barium sulfate particles, creating a novel pigment with enhanced optical and functional properties for architectural and industrial coatings [].
Q10: What are the environmental concerns associated with barium sulfate?
A10: While barium sulfate itself is considered relatively inert and non-toxic, its production and disposal can have environmental impacts. The mining of barite, the primary source of barium sulfate, can lead to habitat destruction and pollution. The responsible management of barium sulfate waste, including recycling and proper disposal practices, is crucial for minimizing environmental harm [].
Q11: How is barium sulfate typically synthesized?
A11: Barium sulfate is commonly synthesized through precipitation reactions involving soluble barium salts, such as barium chloride, and sulfate-containing solutions, like sulfuric acid or sodium sulfate [, ]. Controlling reaction parameters like concentration, temperature, and the presence of additives can influence the particle size, morphology, and purity of the synthesized barium sulfate.
Q12: Are there methods for analyzing the sulfur isotope ratio in barium sulfate derived from historical artifacts containing vermilion (mercuric sulfide)?
A12: Yes, a method for analyzing the sulfur isotope ratio in barium sulfate prepared from vermilion has been developed to prevent mercury contamination []. This method involves dissolving mercuric sulfide in reverse aqua regia, oxidizing sulfur ions to sulfate ions with bromine, and then directly adding barium chloride to precipitate barium sulfate without the need for ion-exchange resins. This technique ensures accurate sulfur isotope analysis while minimizing mercury contamination.
Avertissement et informations sur les produits de recherche in vitro
Veuillez noter que tous les articles et informations sur les produits présentés sur BenchChem sont destinés uniquement à des fins informatives. Les produits disponibles à l'achat sur BenchChem sont spécifiquement conçus pour des études in vitro, qui sont réalisées en dehors des organismes vivants. Les études in vitro, dérivées du terme latin "in verre", impliquent des expériences réalisées dans des environnements de laboratoire contrôlés à l'aide de cellules ou de tissus. Il est important de noter que ces produits ne sont pas classés comme médicaments et n'ont pas reçu l'approbation de la FDA pour la prévention, le traitement ou la guérison de toute condition médicale, affection ou maladie. Nous devons souligner que toute forme d'introduction corporelle de ces produits chez les humains ou les animaux est strictement interdite par la loi. Il est essentiel de respecter ces directives pour assurer la conformité aux normes légales et éthiques en matière de recherche et d'expérimentation.
![[4-[[4-(dimethylamino)phenyl]-(4-dimethylazaniumylidenecyclohexa-2,5-dien-1-ylidene)methyl]phenyl]-ethyl-dimethylazanium;platinum(2+);tetrachloride](/img/structure/B147853.png)
![7-Methyl-2,3-dihydro-1H-benzo[d]pyrrolo[1,2-a]imidazol-5-amine](/img/structure/B147861.png)