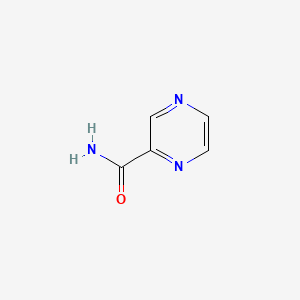
Pyrazinamide
Vue d'ensemble
Description
Le pyrazinamide est un agent antimicrobien principalement utilisé dans le traitement de la tuberculose active. Il est particulièrement efficace lors de la phase initiale du traitement, généralement les deux premiers mois, et est utilisé en association avec d'autres agents antituberculeux. Le this compound présente une activité antibactérienne significative contre Mycobacterium tuberculosis et Mycobacterium africanum .
Mécanisme D'action
Target of Action
Pyrazinamide is a prodrug that is primarily active against Mycobacterium tuberculosis . The primary targets of this compound are the pyrazinamidase (PncA) enzyme, the ribosomal protein S1 (RpsA) , and the aspartate decarboxylase (PanD) . These targets play crucial roles in the survival and replication of the bacteria.
Mode of Action
This compound diffuses into active M. tuberculosis that express the pyrazinamidase enzyme, which converts this compound to its active form, pyrazinoic acid (POA) . POA then interferes with the bacterium’s ability to synthesize new fatty acids, which are required for growth and replication . It has been suggested that POA inhibits the function of the ribosomal protein S1 (RpsA) and the aspartate decarboxylase (PanD), thereby affecting the synthesis of essential metabolic cofactors .
Biochemical Pathways
The biochemical pathways affected by this compound are primarily related to fatty acid synthesis and the synthesis of essential metabolic cofactors . By inhibiting these pathways, this compound disrupts the normal functioning of the bacteria, leading to their eventual death .
Pharmacokinetics
This compound is well absorbed orally, with a bioavailability of over 90% . It is metabolized in the liver and has an elimination half-life of 9 to 10 hours . The drug is excreted through the kidneys . These properties impact the drug’s bioavailability and determine the effective concentration of the drug in the body.
Result of Action
The result of this compound’s action is the inhibition of the growth of M. tuberculosis, leading to the death of the bacteria . This makes this compound a highly specific and effective agent in the treatment of tuberculosis .
Action Environment
This compound is active only at a slightly acidic pH, both in vitro and in vivo . This suggests that the drug’s action, efficacy, and stability are influenced by the environmental pH. This property is particularly relevant in the context of tuberculosis, as the bacteria often reside in slightly acidic environments within the host .
Applications De Recherche Scientifique
Pyrazinamide has a wide range of applications in scientific research:
Chemistry: It is used as a precursor in the synthesis of various pyrazine derivatives.
Biology: this compound is studied for its effects on bacterial cell walls and its role in inhibiting fatty acid synthesis.
Medicine: It is a crucial component in the treatment of tuberculosis, particularly in combination therapies.
Industry: this compound derivatives are explored for their potential use in agricultural chemicals and other industrial applications
Analyse Biochimique
Biochemical Properties
Pyrazinamide is a prodrug that is converted into its active form, pyrazinoic acid, by the enzyme pyrazinamidase, which is expressed in active Mycobacterium tuberculosis . The conversion of this compound to pyrazinoic acid is a critical step in its biochemical activity .
Cellular Effects
This compound has a unique sterilizing effect, killing nonreplicating persisters that other TB drugs fail to kill . This makes it an essential drug for inclusion in any drug combinations for treating TB . It acts differently from common antibiotics by inhibiting multiple targets such as energy production, trans-translation, and perhaps pantothenate/coenzyme A required for persister survival .
Molecular Mechanism
The active form of this compound, pyrazinoic acid, exerts its effects at the molecular level through various mechanisms. It inhibits energy production, disrupts trans-translation, and may also affect the pantothenate/coenzyme A pathway, which is essential for the survival of persisters . Resistance to this compound is primarily driven by genetic variation in the pncA gene, which encodes the enzyme that converts this compound into pyrazinoic acid .
Temporal Effects in Laboratory Settings
The effects of this compound have been extensively studied in laboratory settings. It has been found to have high sterilizing activity, particularly in combination with other TB drugs
Metabolic Pathways
This compound is involved in the metabolic pathways of Mycobacterium tuberculosis. The pncA gene product, pyrazinamidase, is involved in the conversion of the prodrug this compound to its active form, pyrazinoic acid .
Transport and Distribution
This compound diffuses into active M. tuberculosis that express the pyrazinamidase enzyme. The active form, pyrazinoic acid, can leak out under acidic conditions to be converted to the protonated conjugate acid, which readily diffuses back into the bacilli and accumulates intracellularly .
Méthodes De Préparation
Voies de synthèse et conditions de réaction : Le pyrazinamide peut être synthétisé par plusieurs méthodes :
Hydratation de la 2-cyanopyrazine : Cette méthode implique l'hydratation de la 2-cyanopyrazine pour produire du this compound.
Formation de la 2-pyrazinecarboxamide : Une autre méthode implique la réaction de la pyrazine avec la formamide en présence de peroxyde d'hydrogène et de sulfate ferreux pour produire du this compound.
Ammonolyse du 2-pyrazinecarboxylate : Cette méthode utilise le 2-pyrazinecarboxylate, qui subit une ammonolyse en présence d'ammoniac pour former du this compound.
Méthodes de production industrielle : La production industrielle du this compound repose principalement sur l'hydratation de la 2-cyanopyrazine en raison de son rendement élevé et de sa capacité d'adaptation à grande échelle. Cette méthode implique l'utilisation de catalyseurs et de conditions de réaction contrôlées pour garantir une pureté et une efficacité élevées .
Types de réactions :
Oxydation : Le this compound peut subir des réactions d'oxydation, en particulier en présence d'agents oxydants forts.
Réduction : Il peut être réduit en son dérivé amine correspondant dans des conditions spécifiques.
Substitution : Le this compound peut participer à des réactions de substitution, où le groupe amide peut être remplacé par d'autres groupes fonctionnels.
Réactifs et conditions courants :
Agents oxydants : Peroxyde d'hydrogène, permanganate de potassium.
Agents réducteurs : Borohydrure de sodium, hydrure de lithium et d'aluminium.
Réactifs de substitution : Divers agents alkylants et nucléophiles.
Principaux produits :
Produits d'oxydation : Acide pyrazine-2-carboxylique.
Produits de réduction : Pyrazine-2-carboxamide.
Produits de substitution : Divers dérivés pyraziniques substitués.
4. Applications de la recherche scientifique
Le this compound a un large éventail d'applications dans la recherche scientifique :
Chimie : Il est utilisé comme précurseur dans la synthèse de divers dérivés de la pyrazine.
Biologie : Le this compound est étudié pour ses effets sur les parois cellulaires bactériennes et son rôle dans l'inhibition de la synthèse des acides gras.
Médecine : Il est un élément essentiel dans le traitement de la tuberculose, en particulier dans les thérapies combinées.
Industrie : Les dérivés du this compound sont explorés pour leur utilisation potentielle dans les produits chimiques agricoles et autres applications industrielles
5. Mécanisme d'action
Le this compound est un promédicament qui est converti en sa forme active, l'acide pyrazinoïque, par l'enzyme pyrazinamidase dans la cellule bactérienne. L'acide pyrazinoïque perturbe le potentiel de la membrane cellulaire bactérienne et interfère avec la synthèse des acides gras, ce qui conduit à la mort de Mycobacterium tuberculosis. Le médicament est plus actif à des niveaux de pH acides, qui se trouvent généralement dans les macrophages où les bactéries résident .
Composés similaires :
Isoniazide : Un autre agent antituberculeux de première ligne qui inhibe la synthèse des acides mycoliques, des composants essentiels de la paroi cellulaire bactérienne.
Rifampicine : Un antibiotique à large spectre qui inhibe la synthèse de l'ARN bactérien.
Ethambutol : Inhibe la synthèse de l'arabinogalactane, un composant de la paroi cellulaire mycobactérienne.
Unicité du this compound : Le this compound est unique en son genre par sa capacité à cibler Mycobacterium tuberculosis dans des environnements acides, comme ceux qui se trouvent dans les macrophages. Cette propriété lui permet d'être efficace contre les populations bactériennes en cours de multiplication et dormantes, ce qui en fait un élément essentiel de la thérapie de courte durée de la tuberculose .
Comparaison Avec Des Composés Similaires
Isoniazid: Another first-line antituberculous agent that inhibits the synthesis of mycolic acids, essential components of the bacterial cell wall.
Rifampicin: A broad-spectrum antibiotic that inhibits bacterial RNA synthesis.
Ethambutol: Inhibits the synthesis of arabinogalactan, a component of the mycobacterial cell wall.
Uniqueness of Pyrazinamide: this compound is unique in its ability to target Mycobacterium tuberculosis within acidic environments, such as those found in macrophages. This property allows it to be effective against both replicating and dormant bacterial populations, making it a critical component of short-course tuberculosis therapy .
Propriétés
IUPAC Name |
pyrazine-2-carboxamide | |
|---|---|---|
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
InChI |
InChI=1S/C5H5N3O/c6-5(9)4-3-7-1-2-8-4/h1-3H,(H2,6,9) | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
InChI Key |
IPEHBUMCGVEMRF-UHFFFAOYSA-N | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
Canonical SMILES |
C1=CN=C(C=N1)C(=O)N | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
Molecular Formula |
C5H5N3O | |
| Record name | PYRAZINAMIDE | |
| Source | CAMEO Chemicals | |
| URL | https://cameochemicals.noaa.gov/chemical/20970 | |
| Description | CAMEO Chemicals is a chemical database designed for people who are involved in hazardous material incident response and planning. CAMEO Chemicals contains a library with thousands of datasheets containing response-related information and recommendations for hazardous materials that are commonly transported, used, or stored in the United States. CAMEO Chemicals was developed by the National Oceanic and Atmospheric Administration's Office of Response and Restoration in partnership with the Environmental Protection Agency's Office of Emergency Management. | |
| Explanation | CAMEO Chemicals and all other CAMEO products are available at no charge to those organizations and individuals (recipients) responsible for the safe handling of chemicals. However, some of the chemical data itself is subject to the copyright restrictions of the companies or organizations that provided the data. | |
| Record name | pyrazinamide | |
| Source | Wikipedia | |
| URL | https://en.wikipedia.org/wiki/Pyrazinamide | |
| Description | Chemical information link to Wikipedia. | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
DSSTOX Substance ID |
DTXSID9021215 | |
| Record name | Pyrazinamide | |
| Source | EPA DSSTox | |
| URL | https://comptox.epa.gov/dashboard/DTXSID9021215 | |
| Description | DSSTox provides a high quality public chemistry resource for supporting improved predictive toxicology. | |
Molecular Weight |
123.11 g/mol | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
Physical Description |
Pyrazinamide is a white powder. Sublimes from 318 °F. (NTP, 1992), Solid | |
| Record name | PYRAZINAMIDE | |
| Source | CAMEO Chemicals | |
| URL | https://cameochemicals.noaa.gov/chemical/20970 | |
| Description | CAMEO Chemicals is a chemical database designed for people who are involved in hazardous material incident response and planning. CAMEO Chemicals contains a library with thousands of datasheets containing response-related information and recommendations for hazardous materials that are commonly transported, used, or stored in the United States. CAMEO Chemicals was developed by the National Oceanic and Atmospheric Administration's Office of Response and Restoration in partnership with the Environmental Protection Agency's Office of Emergency Management. | |
| Explanation | CAMEO Chemicals and all other CAMEO products are available at no charge to those organizations and individuals (recipients) responsible for the safe handling of chemicals. However, some of the chemical data itself is subject to the copyright restrictions of the companies or organizations that provided the data. | |
| Record name | Pyrazinamide | |
| Source | Human Metabolome Database (HMDB) | |
| URL | http://www.hmdb.ca/metabolites/HMDB0014483 | |
| Description | The Human Metabolome Database (HMDB) is a freely available electronic database containing detailed information about small molecule metabolites found in the human body. | |
| Explanation | HMDB is offered to the public as a freely available resource. Use and re-distribution of the data, in whole or in part, for commercial purposes requires explicit permission of the authors and explicit acknowledgment of the source material (HMDB) and the original publication (see the HMDB citing page). We ask that users who download significant portions of the database cite the HMDB paper in any resulting publications. | |
Boiling Point |
SUB (NTP, 1992) | |
| Record name | PYRAZINAMIDE | |
| Source | CAMEO Chemicals | |
| URL | https://cameochemicals.noaa.gov/chemical/20970 | |
| Description | CAMEO Chemicals is a chemical database designed for people who are involved in hazardous material incident response and planning. CAMEO Chemicals contains a library with thousands of datasheets containing response-related information and recommendations for hazardous materials that are commonly transported, used, or stored in the United States. CAMEO Chemicals was developed by the National Oceanic and Atmospheric Administration's Office of Response and Restoration in partnership with the Environmental Protection Agency's Office of Emergency Management. | |
| Explanation | CAMEO Chemicals and all other CAMEO products are available at no charge to those organizations and individuals (recipients) responsible for the safe handling of chemicals. However, some of the chemical data itself is subject to the copyright restrictions of the companies or organizations that provided the data. | |
Solubility |
>18.5 [ug/mL] (The mean of the results at pH 7.4), Soluble (NTP, 1992), Solubility (mg/ml, 25 °C): methanol 13.8; absolute ethanol 5.7; isopropanol 3.8; ether 1.0; isooctane 0.01; chloroform 7.4, In water, 15 mg/ml @ 25 °C, 9.37e+01 g/L | |
| Record name | SID8139959 | |
| Source | Burnham Center for Chemical Genomics | |
| URL | https://pubchem.ncbi.nlm.nih.gov/bioassay/1996#section=Data-Table | |
| Description | Aqueous solubility in buffer at pH 7.4 | |
| Record name | PYRAZINAMIDE | |
| Source | CAMEO Chemicals | |
| URL | https://cameochemicals.noaa.gov/chemical/20970 | |
| Description | CAMEO Chemicals is a chemical database designed for people who are involved in hazardous material incident response and planning. CAMEO Chemicals contains a library with thousands of datasheets containing response-related information and recommendations for hazardous materials that are commonly transported, used, or stored in the United States. CAMEO Chemicals was developed by the National Oceanic and Atmospheric Administration's Office of Response and Restoration in partnership with the Environmental Protection Agency's Office of Emergency Management. | |
| Explanation | CAMEO Chemicals and all other CAMEO products are available at no charge to those organizations and individuals (recipients) responsible for the safe handling of chemicals. However, some of the chemical data itself is subject to the copyright restrictions of the companies or organizations that provided the data. | |
| Record name | Pyrazinamide | |
| Source | DrugBank | |
| URL | https://www.drugbank.ca/drugs/DB00339 | |
| Description | The DrugBank database is a unique bioinformatics and cheminformatics resource that combines detailed drug (i.e. chemical, pharmacological and pharmaceutical) data with comprehensive drug target (i.e. sequence, structure, and pathway) information. | |
| Explanation | Creative Common's Attribution-NonCommercial 4.0 International License (http://creativecommons.org/licenses/by-nc/4.0/legalcode) | |
| Record name | PYRAZINAMIDE | |
| Source | Hazardous Substances Data Bank (HSDB) | |
| URL | https://pubchem.ncbi.nlm.nih.gov/source/hsdb/3576 | |
| Description | The Hazardous Substances Data Bank (HSDB) is a toxicology database that focuses on the toxicology of potentially hazardous chemicals. It provides information on human exposure, industrial hygiene, emergency handling procedures, environmental fate, regulatory requirements, nanomaterials, and related areas. The information in HSDB has been assessed by a Scientific Review Panel. | |
| Record name | Pyrazinamide | |
| Source | Human Metabolome Database (HMDB) | |
| URL | http://www.hmdb.ca/metabolites/HMDB0014483 | |
| Description | The Human Metabolome Database (HMDB) is a freely available electronic database containing detailed information about small molecule metabolites found in the human body. | |
| Explanation | HMDB is offered to the public as a freely available resource. Use and re-distribution of the data, in whole or in part, for commercial purposes requires explicit permission of the authors and explicit acknowledgment of the source material (HMDB) and the original publication (see the HMDB citing page). We ask that users who download significant portions of the database cite the HMDB paper in any resulting publications. | |
Mechanism of Action |
Pyrazinamide diffuses into active _M. tuberculosis_ that express pyrazinamidase enzyme that converts pyrazinamide to the active form pyrazinoic acid. Pyrazinoic acid can leak out under acidic conditions to be converted to the protonated conjugate acid, which is readily diffused back into the bacilli and accumulate intracellularly. The net effect is that more pyrazinoic acid accumulates inside the bacillus at acid pH than at neutral pH. Pyrazinoic acid was thought to inhibit the enzyme fatty acid synthase (FAS) I, which is required by the bacterium to synthesise fatty acids. However, this theory was thought to have been discounted. However, further studies reproduced the results of FAS I inhibition as the putative mechanism first in whole cell assay of replicating M. tuberculosis bacilli which have shown that pyrazinoic acid and its ester inhibit the synthesis of fatty acids. This study was followed by in vitro assay of tuberculous FAS I enzyme that tested the activity with pyrazinamide, pyrazinoic acid and several classes of pyrazinamide analogs. Pyrazinamide and its analogs inhibited the activity of purified FAS I. It has also been suggested that the accumulation of pyrazinoic acid disrupts membrane potential and interferes with energy production, necessary for survival of M. tuberculosis at an acidic site of infection. Pyrazinoic acid has also been shown to bind to the ribosomal protein S1 (RpsA) and inhibit trans-translation. This may explain the ability of the drug to kill dormant mycobacteria., Pyrazinamide may be bacteriostatic or bactericidal in action, depending on the concentration of the drug attained at the site of the infection and the susceptibility of the infecting organism. In vitro and in vivo, the drug is active only at a slightly acidic pH. The exact mechanism of action of pyrazinamide has not been fully elucidated. The antimycobacterial activity of pyrazinamide appears to partly depend on conversion of the drug to pyrazinoic acid. Susceptible strains of Mycobacterium tuberculosis produce pyrazinamidase, an enzyme that deaminates pyrazinamide to pyrazinoic acid, and the in vitro susceptibility of a given strain of the organism appears to correspond to its pyrazinamidase activity. In vitro studies indicate that pyrazinoic acid has specific antimycobacterial activity against Mycobacterium tuberculosis. In addition, the fact that pyrazinoic acid lowers the pH of the environment below that which is necessary for growth of Mycobacterium tuberculosis appears to contribute to the drug's antimycobacterial activity in vitro., Unknown; pyrazinamide may be bacteriostatic or bactericidal, depending on its concentration and the susceptibility of the organism. It is active in vitro at an acidic pH of 5.6 or less, similar to that found in early, active tubercular inflammatory lesions. | |
| Record name | Pyrazinamide | |
| Source | DrugBank | |
| URL | https://www.drugbank.ca/drugs/DB00339 | |
| Description | The DrugBank database is a unique bioinformatics and cheminformatics resource that combines detailed drug (i.e. chemical, pharmacological and pharmaceutical) data with comprehensive drug target (i.e. sequence, structure, and pathway) information. | |
| Explanation | Creative Common's Attribution-NonCommercial 4.0 International License (http://creativecommons.org/licenses/by-nc/4.0/legalcode) | |
| Record name | PYRAZINAMIDE | |
| Source | Hazardous Substances Data Bank (HSDB) | |
| URL | https://pubchem.ncbi.nlm.nih.gov/source/hsdb/3576 | |
| Description | The Hazardous Substances Data Bank (HSDB) is a toxicology database that focuses on the toxicology of potentially hazardous chemicals. It provides information on human exposure, industrial hygiene, emergency handling procedures, environmental fate, regulatory requirements, nanomaterials, and related areas. The information in HSDB has been assessed by a Scientific Review Panel. | |
Color/Form |
Crystals, Crystals from water or alcohol, Crystals from water or ethyl alcohol | |
CAS No. |
98-96-4 | |
| Record name | PYRAZINAMIDE | |
| Source | CAMEO Chemicals | |
| URL | https://cameochemicals.noaa.gov/chemical/20970 | |
| Description | CAMEO Chemicals is a chemical database designed for people who are involved in hazardous material incident response and planning. CAMEO Chemicals contains a library with thousands of datasheets containing response-related information and recommendations for hazardous materials that are commonly transported, used, or stored in the United States. CAMEO Chemicals was developed by the National Oceanic and Atmospheric Administration's Office of Response and Restoration in partnership with the Environmental Protection Agency's Office of Emergency Management. | |
| Explanation | CAMEO Chemicals and all other CAMEO products are available at no charge to those organizations and individuals (recipients) responsible for the safe handling of chemicals. However, some of the chemical data itself is subject to the copyright restrictions of the companies or organizations that provided the data. | |
| Record name | Pyrazinamide | |
| Source | CAS Common Chemistry | |
| URL | https://commonchemistry.cas.org/detail?cas_rn=98-96-4 | |
| Description | CAS Common Chemistry is an open community resource for accessing chemical information. Nearly 500,000 chemical substances from CAS REGISTRY cover areas of community interest, including common and frequently regulated chemicals, and those relevant to high school and undergraduate chemistry classes. This chemical information, curated by our expert scientists, is provided in alignment with our mission as a division of the American Chemical Society. | |
| Explanation | The data from CAS Common Chemistry is provided under a CC-BY-NC 4.0 license, unless otherwise stated. | |
| Record name | Pyrazinamide [USP:INN:BAN:JAN] | |
| Source | ChemIDplus | |
| URL | https://pubchem.ncbi.nlm.nih.gov/substance/?source=chemidplus&sourceid=0000098964 | |
| Description | ChemIDplus is a free, web search system that provides access to the structure and nomenclature authority files used for the identification of chemical substances cited in National Library of Medicine (NLM) databases, including the TOXNET system. | |
| Record name | Pyrazinamide | |
| Source | DrugBank | |
| URL | https://www.drugbank.ca/drugs/DB00339 | |
| Description | The DrugBank database is a unique bioinformatics and cheminformatics resource that combines detailed drug (i.e. chemical, pharmacological and pharmaceutical) data with comprehensive drug target (i.e. sequence, structure, and pathway) information. | |
| Explanation | Creative Common's Attribution-NonCommercial 4.0 International License (http://creativecommons.org/licenses/by-nc/4.0/legalcode) | |
| Record name | pyrazinamide | |
| Source | DTP/NCI | |
| URL | https://dtp.cancer.gov/dtpstandard/servlet/dwindex?searchtype=NSC&outputformat=html&searchlist=757304 | |
| Description | The NCI Development Therapeutics Program (DTP) provides services and resources to the academic and private-sector research communities worldwide to facilitate the discovery and development of new cancer therapeutic agents. | |
| Explanation | Unless otherwise indicated, all text within NCI products is free of copyright and may be reused without our permission. Credit the National Cancer Institute as the source. | |
| Record name | pyrazinamide | |
| Source | DTP/NCI | |
| URL | https://dtp.cancer.gov/dtpstandard/servlet/dwindex?searchtype=NSC&outputformat=html&searchlist=14911 | |
| Description | The NCI Development Therapeutics Program (DTP) provides services and resources to the academic and private-sector research communities worldwide to facilitate the discovery and development of new cancer therapeutic agents. | |
| Explanation | Unless otherwise indicated, all text within NCI products is free of copyright and may be reused without our permission. Credit the National Cancer Institute as the source. | |
| Record name | 2-Pyrazinecarboxamide | |
| Source | EPA Chemicals under the TSCA | |
| URL | https://www.epa.gov/chemicals-under-tsca | |
| Description | EPA Chemicals under the Toxic Substances Control Act (TSCA) collection contains information on chemicals and their regulations under TSCA, including non-confidential content from the TSCA Chemical Substance Inventory and Chemical Data Reporting. | |
| Record name | Pyrazinamide | |
| Source | EPA DSSTox | |
| URL | https://comptox.epa.gov/dashboard/DTXSID9021215 | |
| Description | DSSTox provides a high quality public chemistry resource for supporting improved predictive toxicology. | |
| Record name | Pyrazinamide | |
| Source | European Chemicals Agency (ECHA) | |
| URL | https://echa.europa.eu/substance-information/-/substanceinfo/100.002.470 | |
| Description | The European Chemicals Agency (ECHA) is an agency of the European Union which is the driving force among regulatory authorities in implementing the EU's groundbreaking chemicals legislation for the benefit of human health and the environment as well as for innovation and competitiveness. | |
| Explanation | Use of the information, documents and data from the ECHA website is subject to the terms and conditions of this Legal Notice, and subject to other binding limitations provided for under applicable law, the information, documents and data made available on the ECHA website may be reproduced, distributed and/or used, totally or in part, for non-commercial purposes provided that ECHA is acknowledged as the source: "Source: European Chemicals Agency, http://echa.europa.eu/". Such acknowledgement must be included in each copy of the material. ECHA permits and encourages organisations and individuals to create links to the ECHA website under the following cumulative conditions: Links can only be made to webpages that provide a link to the Legal Notice page. | |
| Record name | PYRAZINAMIDE | |
| Source | FDA Global Substance Registration System (GSRS) | |
| URL | https://gsrs.ncats.nih.gov/ginas/app/beta/substances/2KNI5N06TI | |
| Description | The FDA Global Substance Registration System (GSRS) enables the efficient and accurate exchange of information on what substances are in regulated products. Instead of relying on names, which vary across regulatory domains, countries, and regions, the GSRS knowledge base makes it possible for substances to be defined by standardized, scientific descriptions. | |
| Explanation | Unless otherwise noted, the contents of the FDA website (www.fda.gov), both text and graphics, are not copyrighted. They are in the public domain and may be republished, reprinted and otherwise used freely by anyone without the need to obtain permission from FDA. Credit to the U.S. Food and Drug Administration as the source is appreciated but not required. | |
| Record name | PYRAZINAMIDE | |
| Source | Hazardous Substances Data Bank (HSDB) | |
| URL | https://pubchem.ncbi.nlm.nih.gov/source/hsdb/3576 | |
| Description | The Hazardous Substances Data Bank (HSDB) is a toxicology database that focuses on the toxicology of potentially hazardous chemicals. It provides information on human exposure, industrial hygiene, emergency handling procedures, environmental fate, regulatory requirements, nanomaterials, and related areas. The information in HSDB has been assessed by a Scientific Review Panel. | |
| Record name | Pyrazinamide | |
| Source | Human Metabolome Database (HMDB) | |
| URL | http://www.hmdb.ca/metabolites/HMDB0014483 | |
| Description | The Human Metabolome Database (HMDB) is a freely available electronic database containing detailed information about small molecule metabolites found in the human body. | |
| Explanation | HMDB is offered to the public as a freely available resource. Use and re-distribution of the data, in whole or in part, for commercial purposes requires explicit permission of the authors and explicit acknowledgment of the source material (HMDB) and the original publication (see the HMDB citing page). We ask that users who download significant portions of the database cite the HMDB paper in any resulting publications. | |
Melting Point |
376 to 379 °F (NTP, 1992), 192 °C | |
| Record name | PYRAZINAMIDE | |
| Source | CAMEO Chemicals | |
| URL | https://cameochemicals.noaa.gov/chemical/20970 | |
| Description | CAMEO Chemicals is a chemical database designed for people who are involved in hazardous material incident response and planning. CAMEO Chemicals contains a library with thousands of datasheets containing response-related information and recommendations for hazardous materials that are commonly transported, used, or stored in the United States. CAMEO Chemicals was developed by the National Oceanic and Atmospheric Administration's Office of Response and Restoration in partnership with the Environmental Protection Agency's Office of Emergency Management. | |
| Explanation | CAMEO Chemicals and all other CAMEO products are available at no charge to those organizations and individuals (recipients) responsible for the safe handling of chemicals. However, some of the chemical data itself is subject to the copyright restrictions of the companies or organizations that provided the data. | |
| Record name | Pyrazinamide | |
| Source | DrugBank | |
| URL | https://www.drugbank.ca/drugs/DB00339 | |
| Description | The DrugBank database is a unique bioinformatics and cheminformatics resource that combines detailed drug (i.e. chemical, pharmacological and pharmaceutical) data with comprehensive drug target (i.e. sequence, structure, and pathway) information. | |
| Explanation | Creative Common's Attribution-NonCommercial 4.0 International License (http://creativecommons.org/licenses/by-nc/4.0/legalcode) | |
| Record name | PYRAZINAMIDE | |
| Source | Hazardous Substances Data Bank (HSDB) | |
| URL | https://pubchem.ncbi.nlm.nih.gov/source/hsdb/3576 | |
| Description | The Hazardous Substances Data Bank (HSDB) is a toxicology database that focuses on the toxicology of potentially hazardous chemicals. It provides information on human exposure, industrial hygiene, emergency handling procedures, environmental fate, regulatory requirements, nanomaterials, and related areas. The information in HSDB has been assessed by a Scientific Review Panel. | |
| Record name | Pyrazinamide | |
| Source | Human Metabolome Database (HMDB) | |
| URL | http://www.hmdb.ca/metabolites/HMDB0014483 | |
| Description | The Human Metabolome Database (HMDB) is a freely available electronic database containing detailed information about small molecule metabolites found in the human body. | |
| Explanation | HMDB is offered to the public as a freely available resource. Use and re-distribution of the data, in whole or in part, for commercial purposes requires explicit permission of the authors and explicit acknowledgment of the source material (HMDB) and the original publication (see the HMDB citing page). We ask that users who download significant portions of the database cite the HMDB paper in any resulting publications. | |
Synthesis routes and methods I
Procedure details








Synthesis routes and methods II
Procedure details











Synthesis routes and methods III
Procedure details








Retrosynthesis Analysis
AI-Powered Synthesis Planning: Our tool employs the Template_relevance Pistachio, Template_relevance Bkms_metabolic, Template_relevance Pistachio_ringbreaker, Template_relevance Reaxys, Template_relevance Reaxys_biocatalysis model, leveraging a vast database of chemical reactions to predict feasible synthetic routes.
One-Step Synthesis Focus: Specifically designed for one-step synthesis, it provides concise and direct routes for your target compounds, streamlining the synthesis process.
Accurate Predictions: Utilizing the extensive PISTACHIO, BKMS_METABOLIC, PISTACHIO_RINGBREAKER, REAXYS, REAXYS_BIOCATALYSIS database, our tool offers high-accuracy predictions, reflecting the latest in chemical research and data.
Strategy Settings
| Precursor scoring | Relevance Heuristic |
|---|---|
| Min. plausibility | 0.01 |
| Model | Template_relevance |
| Template Set | Pistachio/Bkms_metabolic/Pistachio_ringbreaker/Reaxys/Reaxys_biocatalysis |
| Top-N result to add to graph | 6 |
Feasible Synthetic Routes
Avertissement et informations sur les produits de recherche in vitro
Veuillez noter que tous les articles et informations sur les produits présentés sur BenchChem sont destinés uniquement à des fins informatives. Les produits disponibles à l'achat sur BenchChem sont spécifiquement conçus pour des études in vitro, qui sont réalisées en dehors des organismes vivants. Les études in vitro, dérivées du terme latin "in verre", impliquent des expériences réalisées dans des environnements de laboratoire contrôlés à l'aide de cellules ou de tissus. Il est important de noter que ces produits ne sont pas classés comme médicaments et n'ont pas reçu l'approbation de la FDA pour la prévention, le traitement ou la guérison de toute condition médicale, affection ou maladie. Nous devons souligner que toute forme d'introduction corporelle de ces produits chez les humains ou les animaux est strictement interdite par la loi. Il est essentiel de respecter ces directives pour assurer la conformité aux normes légales et éthiques en matière de recherche et d'expérimentation.
![(2E,4E)-5-[(1S,7S,8S,9R)-7-acetyloxy-4-methoxycarbonyl-9-methyl-11-oxo-10-oxatricyclo[6.3.2.01,7]tridec-3-en-9-yl]-2-methylpenta-2,4-dienoic acid](/img/structure/B1679821.png)
![(2'S,3S,4aR,7R,8R,8aR)-8-[(1R)-1-acetyloxy-2-carboxyethyl]-3-[(S)-furan-3-yl-[(2R,3R,4S,5S,6R)-3,4,5-trihydroxy-6-(hydroxymethyl)oxan-2-yl]oxymethyl]-7-(2-hydroxypropan-2-yl)-3,4a,8-trimethyl-5-oxospiro[2,6,7,8a-tetrahydro-1H-naphthalene-4,3'-oxirane]-2'-carboxylic acid](/img/structure/B1679834.png)