Osimertinib D6
概要
説明
Osimertinib D6 is a deuterated form of osimertinib, a third-generation epidermal growth factor receptor tyrosine kinase inhibitor. It is primarily used in the treatment of non-small cell lung cancer with specific mutations in the epidermal growth factor receptor gene. The deuterated version, this compound, is designed to improve the pharmacokinetic properties of the original compound by replacing certain hydrogen atoms with deuterium.
作用機序
Target of Action
Osimertinib D6, also known as Osimertinib-d6, is a deuterated compound of Osimertinib . Osimertinib is a third-generation epidermal growth factor receptor (EGFR) tyrosine kinase inhibitor (TKI) developed by AstraZeneca Pharmaceuticals . It is primarily targeted at certain mutant forms of EGFR (T790M, L858R, and exon 19 deletion) that predominate in non-small cell lung cancer (NSCLC) tumors following treatment with first-line EGFR-TKIs .
Mode of Action
Osimertinib binds to the EGFR mutants, inhibiting their tyrosine kinase activity . This inhibition prevents the phosphorylation and activation of the downstream signaling pathways, thereby inhibiting the growth and proliferation of cancer cells . Osimertinib has been shown to spare wild-type EGFR during therapy, thereby reducing non-specific binding and limiting toxicity .
Biochemical Pathways
Osimertinib primarily affects the EGFR signaling pathway. By inhibiting the tyrosine kinase activity of EGFR mutants, it prevents the activation of downstream signaling pathways involved in cell proliferation, survival, and other cellular functions . The development of third-generation EGFR-TKIs, such as osimertinib, has been in response to altered tumor resistance patterns following treatment .
Pharmacokinetics
Osimertinib is largely excreted in feces (~68%) and to a lesser extent in urine (~14%). Unchanged osimertinib accounted for approximately 2% of the total elimination with 0.8% in urine and 1.2% in feces . The murine Cyp2d gene cluster played a predominant role in mouse, whereas CYP3A4 was the major human enzyme responsible for osimertinib metabolism .
Result of Action
The primary result of Osimertinib’s action is the inhibition of growth and proliferation of NSCLC cells with specific EGFR mutations . This leads to a clinically effective response in approximately 10% of patients with NSCLC . Resistance to osimertinib inevitably develops during the treatment, limiting its long-term effectiveness .
Action Environment
The tumor microenvironment (TME) plays a significant role in the action of Osimertinib. Interactions between the tumor and the TME account for drug resistance . Furthermore, environmental factors such as smoking can influence the action, efficacy, and stability of Osimertinib . For example, a P450 enzyme expressed in smokers’ lungs and lung tumors has the capacity to metabolize Osimertinib .
生化学分析
Biochemical Properties
Osimertinib D6 interacts with several enzymes and proteins. It is a potent inhibitor of EGFR, specifically targeting the L858R and L858R/T790M mutations . It works by forming a covalent bond with the Cys797 residue located in the ATP-binding site of mutant EGFR . This interaction inhibits the ATP-binding cassette (ABC) transporter-mediated multidrug resistance (MDR) in cancer cells .
Cellular Effects
This compound has significant effects on various types of cells and cellular processes. It influences cell function by impacting cell signaling pathways, gene expression, and cellular metabolism . It has been shown to sensitize both ABCB1-transfected and drug-selected cell lines to substrate anticancer drugs . It also significantly increases the accumulation of paclitaxel in ABCB1 overexpressing cells by blocking the efflux function of the ABCB1 transporter .
Molecular Mechanism
This compound exerts its effects at the molecular level through several mechanisms. It forms a covalent bond with the Cys797 residue in the ATP-binding site of mutant EGFR, thereby inhibiting the enzyme . This interaction leads to the inhibition of the ABC transporter-mediated MDR in cancer cells . It also stimulates the ATPase activity of the ABCB1 transporter .
Temporal Effects in Laboratory Settings
The effects of this compound change over time in laboratory settings. While specific data on this compound is limited, studies on Osimertinib have shown that resistance to the drug inevitably develops over time . This resistance is a major challenge in the treatment of EGFR-mutated advanced NSCLC .
Metabolic Pathways
This compound is involved in several metabolic pathways. It is mainly metabolized by the CYP3A enzyme in humans . Species differences in Osimertinib metabolism have been identified, and these can be eliminated by genetic modification .
準備方法
Synthetic Routes and Reaction Conditions: The synthesis of Osimertinib D6 involves the incorporation of deuterium atoms into the osimertinib molecule. This can be achieved through various methods, including:
Hydrogen-Deuterium Exchange: This method involves the replacement of hydrogen atoms with deuterium in the presence of a deuterium source, such as deuterium oxide or deuterated solvents.
Deuterated Reagents: Using deuterated reagents in the synthesis process can directly introduce deuterium atoms into the molecule.
Industrial Production Methods: Industrial production of this compound follows similar principles as the laboratory synthesis but on a larger scale. The process involves:
Large-Scale Hydrogen-Deuterium Exchange: Utilizing deuterium oxide or other deuterated solvents in large quantities.
Optimization of Reaction Conditions: Ensuring the reaction conditions are optimized for maximum yield and purity of the deuterated product.
化学反応の分析
Types of Reactions: Osimertinib D6 undergoes various chemical reactions, including:
Oxidation: The compound can be oxidized to form various metabolites.
Reduction: Reduction reactions can modify the functional groups within the molecule.
Substitution: Substitution reactions can occur at specific sites within the molecule, leading to the formation of different derivatives.
Common Reagents and Conditions:
Oxidation Reagents: Common oxidation reagents include hydrogen peroxide and potassium permanganate.
Reduction Reagents: Sodium borohydride and lithium aluminum hydride are commonly used for reduction reactions.
Substitution Reagents: Halogenating agents and nucleophiles are used for substitution reactions.
Major Products Formed: The major products formed from these reactions include various metabolites and derivatives of this compound, which can have different pharmacological properties.
科学的研究の応用
Osimertinib D6 has several scientific research applications, including:
Chemistry: Used as a model compound to study the effects of deuterium substitution on the pharmacokinetics and pharmacodynamics of drugs.
Biology: Investigated for its interactions with biological targets, such as the epidermal growth factor receptor.
Medicine: Used in preclinical and clinical studies to evaluate its efficacy and safety in treating non-small cell lung cancer.
Industry: Employed in the development of new deuterated drugs with improved properties.
類似化合物との比較
Gefitinib: A first-generation epidermal growth factor receptor tyrosine kinase inhibitor.
Erlotinib: Another first-generation epidermal growth factor receptor tyrosine kinase inhibitor.
Afatinib: A second-generation epidermal growth factor receptor tyrosine kinase inhibitor.
Dacomitinib: Another second-generation epidermal growth factor receptor tyrosine kinase inhibitor.
Comparison: Osimertinib D6 is unique among these compounds due to its third-generation status and its ability to target the T790M resistance mutation. This makes it particularly effective in patients who have developed resistance to first- and second-generation inhibitors. Additionally, the deuterium substitution in this compound can enhance its pharmacokinetic properties, potentially leading to improved efficacy and reduced side effects.
特性
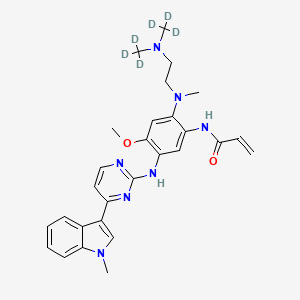
IUPAC Name |
N-[2-[2-[bis(trideuteriomethyl)amino]ethyl-methylamino]-4-methoxy-5-[[4-(1-methylindol-3-yl)pyrimidin-2-yl]amino]phenyl]prop-2-enamide | |
|---|---|---|
| Details | Computed by Lexichem TK 2.7.0 (PubChem release 2021.05.07) | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
InChI |
InChI=1S/C28H33N7O2/c1-7-27(36)30-22-16-23(26(37-6)17-25(22)34(4)15-14-33(2)3)32-28-29-13-12-21(31-28)20-18-35(5)24-11-9-8-10-19(20)24/h7-13,16-18H,1,14-15H2,2-6H3,(H,30,36)(H,29,31,32)/i2D3,3D3 | |
| Details | Computed by InChI 1.0.6 (PubChem release 2021.05.07) | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
InChI Key |
DUYJMQONPNNFPI-XERRXZQWSA-N | |
| Details | Computed by InChI 1.0.6 (PubChem release 2021.05.07) | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
Canonical SMILES |
CN1C=C(C2=CC=CC=C21)C3=NC(=NC=C3)NC4=C(C=C(C(=C4)NC(=O)C=C)N(C)CCN(C)C)OC | |
| Details | Computed by OEChem 2.3.0 (PubChem release 2021.05.07) | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
Isomeric SMILES |
[2H]C([2H])([2H])N(CCN(C)C1=CC(=C(C=C1NC(=O)C=C)NC2=NC=CC(=N2)C3=CN(C4=CC=CC=C43)C)OC)C([2H])([2H])[2H] | |
| Details | Computed by OEChem 2.3.0 (PubChem release 2021.05.07) | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
Molecular Formula |
C28H33N7O2 | |
| Details | Computed by PubChem 2.1 (PubChem release 2021.05.07) | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
Molecular Weight |
505.6 g/mol | |
| Details | Computed by PubChem 2.1 (PubChem release 2021.05.07) | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
Synthesis routes and methods I
Procedure details








Synthesis routes and methods II
Procedure details










Synthesis routes and methods III
Procedure details







試験管内研究製品の免責事項と情報
BenchChemで提示されるすべての記事および製品情報は、情報提供を目的としています。BenchChemで購入可能な製品は、生体外研究のために特別に設計されています。生体外研究は、ラテン語の "in glass" に由来し、生物体の外で行われる実験を指します。これらの製品は医薬品または薬として分類されておらず、FDAから任何の医療状態、病気、または疾患の予防、治療、または治癒のために承認されていません。これらの製品を人間または動物に体内に導入する形態は、法律により厳格に禁止されています。これらのガイドラインに従うことは、研究と実験において法的および倫理的な基準の遵守を確実にするために重要です。
![(4-(Furan-2-carbonyl)piperazin-1-yl)(6-(4-methoxyphenyl)imidazo[2,1-b]thiazol-3-yl)methanone](/img/structure/B2766884.png)
![2-(8-methoxy-6-methyl-10-oxo-3,4,5,10-tetrahydrobenzo[b][1,6]naphthyridin-2(1H)-yl)-N-(2-methoxyphenyl)acetamide](/img/structure/B2766886.png)
![N-(4-chlorobenzo[d]thiazol-2-yl)-4-phenoxy-N-(pyridin-3-ylmethyl)benzamide](/img/structure/B2766887.png)
![(E)-N-(6-methoxy-3-(prop-2-yn-1-yl)benzo[d]thiazol-2(3H)-ylidene)-3-methylisoxazole-5-carboxamide](/img/structure/B2766888.png)
![(E)-N-(6-chloro-3-ethylbenzo[d]thiazol-2(3H)-ylidene)isobutyramide](/img/structure/B2766893.png)
![6-[2-(2-Cyanoanilino)vinyl]pyrazolo[1,5-a]pyrido[3,4-e]pyrimidine-3-carbonitrile](/img/structure/B2766897.png)