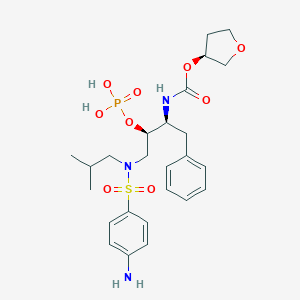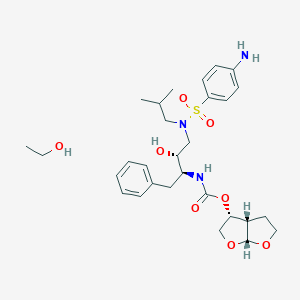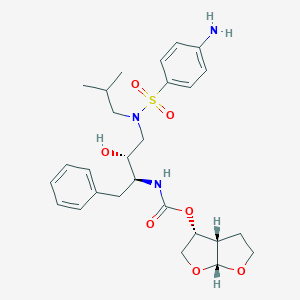
Darunavir
Übersicht
Beschreibung
Darunavir ist ein nicht-peptidischer Proteaseinhibitor, der hauptsächlich zur Behandlung und Vorbeugung von Infektionen mit dem humanen Immundefizienzvirus (HIV) eingesetzt wird. Es wird häufig in Kombination mit anderen antiretroviralen Mitteln verabreicht, um seine Wirksamkeit zu erhöhen. This compound wurde 2006 von der US-amerikanischen Food and Drug Administration zugelassen und ist in der Liste der unentbehrlichen Arzneimittel der Weltgesundheitsorganisation enthalten .
2. Herstellungsmethoden
Synthetische Wege und Reaktionsbedingungen: Die Synthese von this compound umfasst mehrere Schritte, darunter die Bildung von Schlüsselzwischenprodukten und deren anschließende Kupplung. Ein gängiger Syntheseweg umfasst die Entfernung der BOC-Gruppe unter Verwendung von Trifluoressigsäure in Dichlormethan, gefolgt von der Reaktion mit einem Carbonat in Gegenwart von Triethylamin, um this compound zu ergeben .
Industrielle Produktionsmethoden: Die industrielle Produktion von Darunavirethanolat beinhaltet einen robusten Prozess mit mehreren Isolierungs- und Trocknungsschritten. Dieser Prozess stellt eine hohe chemische Ausbeute und Reinheit sicher, wobei kritische Prozessverunreinigungen unter den gewünschten Grenzen gehalten werden . Darüber hinaus werden Verfahren wie Heißschmelzextrusion und Sprühtrocknung eingesetzt, um die Löslichkeit und Bioverfügbarkeit von this compound zu verbessern .
Wirkmechanismus
Target of Action
Darunavir is primarily targeted at the human immunodeficiency virus (HIV) protease . This enzyme plays a crucial role in the life cycle of the HIV virus, making it an attractive target for antiretroviral therapy .
Mode of Action
This compound works by binding to the active site of the HIV-1 protease enzyme , thereby preventing the dimerization and the catalytic activity of the HIV-1 protease . This binding inhibits the necessary viral precursor protein processing and viral maturation in preparation for infection . This compound has a dual mode of action – it binds to the dimeric active site and prevents the dimerization by binding to the HIVPR monomer .
Biochemical Pathways
The primary biochemical pathway affected by this compound is the HIV replication pathway . By inhibiting the HIV-1 protease enzyme, this compound prevents the proper formation of essential proteins and enzymes, which disrupts the assembly and maturation of new viral particles .
Pharmacokinetics
This compound is heavily oxidized and metabolized by hepatic cytochrome enzymes, mainly CYP3A . The bioavailability of this compound is 37% without ritonavir and 82% with ritonavir . The terminal elimination half-life of this compound is 15 hours in the presence of ritonavir .
Result of Action
The result of this compound’s action is a significant decrease in viral load and an increase in CD4 cell counts , which reduces the morbidity and mortality of HIV infection . This leads to the suppression of the HIV virus, preventing it from infecting new cells and helping to control the progression of the disease .
Action Environment
The action of this compound can be influenced by various environmental factors. For instance, the presence of other medications can affect the metabolism of this compound, especially those that are also metabolized by the CYP3A enzyme . Additionally, the presence of ritonavir, a pharmacokinetic enhancer, significantly increases the bioavailability of this compound .
Wissenschaftliche Forschungsanwendungen
Darunavir hat ein breites Spektrum an Anwendungen in der wissenschaftlichen Forschung:
Chemie: Wird als Modellverbindung bei der Untersuchung von Proteaseinhibitoren verwendet.
Biologie: Untersucht wegen seiner Wechselwirkungen mit HIV-Protease und anderen biologischen Zielmolekülen.
Medizin: Wird hauptsächlich zur Behandlung von HIV/AIDS eingesetzt.
5. Wirkmechanismus
This compound übt seine Wirkung aus, indem es das HIV-1-Protease-Enzym hemmt, das für die Verarbeitung von viralen Vorläuferproteinen und die virale Reifung unerlässlich ist. Durch die Bindung an die aktive Stelle des Enzyms verhindert this compound die Spaltung des HIV Gag-Pol-Polyproteins, wodurch die Bildung infektiöser Virionen blockiert wird . Dieser Mechanismus ist entscheidend für seine antiretrovirale Aktivität.
Ähnliche Verbindungen:
- Amprenavir
- Indinavir
- Saquinavir
- Nelfinavir
- Ritonavir
Vergleich: this compound ist unter den Proteaseinhibitoren einzigartig, da es eine hohe genetische Barriere gegen Resistenzen aufweist und eine große Bandbreite an Proteaseinhibitor-resistenten HIV-1-Stämmen hemmen kann . Im Gegensatz zu einigen anderen Proteaseinhibitoren wird this compound oft in Kombination mit Ritonavir oder Cobicistat verwendet, um sein pharmakokinetisches Profil zu verbessern .
Biochemische Analyse
Biochemical Properties
Darunavir plays a crucial role in biochemical reactions by inhibiting the activity of the HIV-1 protease enzyme. This enzyme is responsible for cleaving the viral polyprotein into functional proteins necessary for viral replication . This compound binds to the active site of the HIV-1 protease through multiple hydrogen bonds, thereby preventing the enzyme from processing the viral polyprotein . This inhibition leads to the production of immature, non-infectious viral particles . This compound interacts with various biomolecules, including the HIV-1 protease enzyme and other proteins involved in the viral replication process .
Cellular Effects
This compound has significant effects on various types of cells and cellular processes. In HIV-infected cells, this compound reduces viral load and increases CD4 cell counts, which are crucial for maintaining a healthy immune system . By inhibiting the HIV-1 protease enzyme, this compound disrupts the viral replication cycle, leading to a decrease in the production of new viral particles . This inhibition also affects cell signaling pathways, gene expression, and cellular metabolism by preventing the virus from hijacking the host cell’s machinery .
Molecular Mechanism
The molecular mechanism of this compound involves its binding to the active site of the HIV-1 protease enzyme. This compound forms multiple hydrogen bonds with the enzyme, which stabilizes its binding and prevents the protease from cleaving the viral polyprotein . This inhibition results in the production of immature viral particles that are unable to infect new cells . Additionally, this compound’s high affinity for the protease enzyme makes it effective against HIV strains that have developed resistance to other protease inhibitors .
Temporal Effects in Laboratory Settings
In laboratory settings, the effects of this compound have been observed to change over time. This compound is generally stable and maintains its efficacy over extended periods . Long-term studies have shown that resistance-associated mutations can emerge in patients experiencing virological failure during prolonged use of this compound . These mutations can reduce the drug’s effectiveness, necessitating adjustments in treatment regimens .
Dosage Effects in Animal Models
The effects of this compound vary with different dosages in animal models. Studies have shown that higher doses of this compound result in increased drug concentrations in the brain, liver, and plasma . At high doses, this compound can also cause toxic or adverse effects, such as gastrointestinal disturbances and lipid abnormalities . It is essential to determine the optimal dosage to maximize efficacy while minimizing adverse effects .
Metabolic Pathways
This compound is primarily metabolized by the cytochrome P450 3A (CYP3A) isoenzymes in the liver . The metabolic pathways involve carbamate hydrolysis, isobutyl aliphatic hydroxylation, and aniline aromatic hydroxylation . Ritonavir, a CYP3A inhibitor, is often co-administered with this compound to enhance its bioavailability and prolong its half-life . This combination allows for lower daily doses of this compound while maintaining its therapeutic efficacy .
Transport and Distribution
This compound is transported and distributed within cells and tissues through various mechanisms. It exhibits sufficient membrane permeability to achieve adequate intestinal absorption . The drug is also subject to active transport processes, such as those mediated by P-glycoprotein (P-gp) or other efflux proteins . These transporters play a role in the drug’s localization and accumulation within different tissues .
Subcellular Localization
The subcellular localization of this compound is primarily within the cytoplasm, where it interacts with the HIV-1 protease enzyme . This compound’s activity is not significantly affected by targeting signals or post-translational modifications, as its primary function is to inhibit the protease enzyme within the cytoplasmic compartment . This localization ensures that this compound effectively disrupts the viral replication process within infected cells .
Vorbereitungsmethoden
Synthetic Routes and Reaction Conditions: The synthesis of darunavir involves multiple steps, including the formation of key intermediates and their subsequent coupling. One common synthetic route includes the removal of the BOC group using trifluoroacetic acid in dichloromethane, followed by reaction with a carbonate in the presence of triethylamine to yield this compound .
Industrial Production Methods: Industrial production of this compound ethanolate involves a robust process with multiple isolations and drying steps. This process ensures high chemical yield and purity, with critical process impurities controlled below desired limits . Additionally, methods such as hot-melt extrusion and spray-drying are employed to improve the solubility and bioavailability of this compound .
Analyse Chemischer Reaktionen
Arten von Reaktionen: Darunavir unterliegt verschiedenen chemischen Reaktionen, darunter Oxidation, Reduktion und Substitution. Es wird stark oxidiert und metabolisiert durch hepatische Cytochrom-Enzyme, hauptsächlich CYP3A4 .
Häufige Reagenzien und Bedingungen:
Oxidation: Umfasst hepatische Cytochrom-Enzyme.
Reduktion: Für this compound nicht häufig berichtet.
Substitution: Umfasst Reaktionen mit Carbonaten und Aminen.
Hauptprodukte: Die Hauptprodukte, die aus diesen Reaktionen entstehen, sind hydroxylierte und glucuronidierte Metaboliten .
Vergleich Mit ähnlichen Verbindungen
- Amprenavir
- Indinavir
- Saquinavir
- Nelfinavir
- Ritonavir
Comparison: Darunavir is unique among protease inhibitors due to its high genetic barrier to resistance and its ability to inhibit a wide range of protease inhibitor-resistant HIV-1 strains . Unlike some other protease inhibitors, this compound is often used in combination with ritonavir or cobicistat to enhance its pharmacokinetic profile .
Eigenschaften
IUPAC Name |
[(3aS,4R,6aR)-2,3,3a,4,5,6a-hexahydrofuro[2,3-b]furan-4-yl] N-[(2S,3R)-4-[(4-aminophenyl)sulfonyl-(2-methylpropyl)amino]-3-hydroxy-1-phenylbutan-2-yl]carbamate | |
|---|---|---|
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
InChI |
InChI=1S/C27H37N3O7S/c1-18(2)15-30(38(33,34)21-10-8-20(28)9-11-21)16-24(31)23(14-19-6-4-3-5-7-19)29-27(32)37-25-17-36-26-22(25)12-13-35-26/h3-11,18,22-26,31H,12-17,28H2,1-2H3,(H,29,32)/t22-,23-,24+,25-,26+/m0/s1 | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
InChI Key |
CJBJHOAVZSMMDJ-HEXNFIEUSA-N | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
Canonical SMILES |
CC(C)CN(CC(C(CC1=CC=CC=C1)NC(=O)OC2COC3C2CCO3)O)S(=O)(=O)C4=CC=C(C=C4)N | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
Isomeric SMILES |
CC(C)CN(C[C@H]([C@H](CC1=CC=CC=C1)NC(=O)O[C@H]2CO[C@@H]3[C@H]2CCO3)O)S(=O)(=O)C4=CC=C(C=C4)N | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
Molecular Formula |
C27H37N3O7S | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
DSSTOX Substance ID |
DTXSID0046779 | |
| Record name | Darunavir | |
| Source | EPA DSSTox | |
| URL | https://comptox.epa.gov/dashboard/DTXSID0046779 | |
| Description | DSSTox provides a high quality public chemistry resource for supporting improved predictive toxicology. | |
Molecular Weight |
547.7 g/mol | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
Physical Description |
Solid | |
| Record name | Darunavir | |
| Source | Human Metabolome Database (HMDB) | |
| URL | http://www.hmdb.ca/metabolites/HMDB0015393 | |
| Description | The Human Metabolome Database (HMDB) is a freely available electronic database containing detailed information about small molecule metabolites found in the human body. | |
| Explanation | HMDB is offered to the public as a freely available resource. Use and re-distribution of the data, in whole or in part, for commercial purposes requires explicit permission of the authors and explicit acknowledgment of the source material (HMDB) and the original publication (see the HMDB citing page). We ask that users who download significant portions of the database cite the HMDB paper in any resulting publications. | |
Solubility |
Approximately 0.15 mg/mL at, 6.68e-02 g/L | |
| Record name | Darunavir | |
| Source | DrugBank | |
| URL | https://www.drugbank.ca/drugs/DB01264 | |
| Description | The DrugBank database is a unique bioinformatics and cheminformatics resource that combines detailed drug (i.e. chemical, pharmacological and pharmaceutical) data with comprehensive drug target (i.e. sequence, structure, and pathway) information. | |
| Explanation | Creative Common's Attribution-NonCommercial 4.0 International License (http://creativecommons.org/licenses/by-nc/4.0/legalcode) | |
| Record name | Darunavir | |
| Source | Human Metabolome Database (HMDB) | |
| URL | http://www.hmdb.ca/metabolites/HMDB0015393 | |
| Description | The Human Metabolome Database (HMDB) is a freely available electronic database containing detailed information about small molecule metabolites found in the human body. | |
| Explanation | HMDB is offered to the public as a freely available resource. Use and re-distribution of the data, in whole or in part, for commercial purposes requires explicit permission of the authors and explicit acknowledgment of the source material (HMDB) and the original publication (see the HMDB citing page). We ask that users who download significant portions of the database cite the HMDB paper in any resulting publications. | |
Mechanism of Action |
The HIV-1 protease enzyme is necessary for viral precursor protein processing and viral maturation in preparation for infection, and is therefore a target for antiretroviral therapy for HIV. Protease inhibitors are used as a part of highly active antiretroviral therapy (HAART) in patients diagnosed with HIV infection. It has been shown to effectively suppress the virus, leading to significantly decreased morbidity and mortality rates. Darunavir, a HIV protease inhibitor, prevents HIV replication through binding to the enzyme, stopping the dimerization and the catalytic activity of HIV-1 protease. In particular, it inhibits the cleavage of HIV encoded Gag-Pol proteins in cells that have been infected with the virus, halting the formation of mature virus particles, which spread the infection. The close contact that darunavir makes with the primary chains of the active site amino acids (Asp-29 and Asp-30) on the protease likely contributes to its potency and efficacy against resistant variants of HIV-1. Darunavir is known to bind to different sites on the enzyme: the active site cavity and the surface of one of the flexible flaps in the protease dimer. Darunavir can adapt to changes in the shape of a protease enzyme due to its molecular flexibility., Darunavir as a protease inhibitor inhibits the cleavage of HIV encoded gag-pol polyproteins in virus infected cells, thereby preventing the formation of mature and infectious new virions. It was selected for its potency against wild type HIV-1 and HIV strains resistant to currently approved protease inhibitors., Darunavir is an inhibitor of the HIV-1 protease. It selectively inhibits the cleavage of HIV encoded Gag-Pol polyproteins in infected cells, thereby preventing the formation of mature virus particles. | |
| Record name | Darunavir | |
| Source | DrugBank | |
| URL | https://www.drugbank.ca/drugs/DB01264 | |
| Description | The DrugBank database is a unique bioinformatics and cheminformatics resource that combines detailed drug (i.e. chemical, pharmacological and pharmaceutical) data with comprehensive drug target (i.e. sequence, structure, and pathway) information. | |
| Explanation | Creative Common's Attribution-NonCommercial 4.0 International License (http://creativecommons.org/licenses/by-nc/4.0/legalcode) | |
| Record name | Darunavir | |
| Source | Hazardous Substances Data Bank (HSDB) | |
| URL | https://pubchem.ncbi.nlm.nih.gov/source/hsdb/7788 | |
| Description | The Hazardous Substances Data Bank (HSDB) is a toxicology database that focuses on the toxicology of potentially hazardous chemicals. It provides information on human exposure, industrial hygiene, emergency handling procedures, environmental fate, regulatory requirements, nanomaterials, and related areas. The information in HSDB has been assessed by a Scientific Review Panel. | |
Color/Form |
White, amorphous solid | |
CAS No. |
206361-99-1 | |
| Record name | Darunavir | |
| Source | CAS Common Chemistry | |
| URL | https://commonchemistry.cas.org/detail?cas_rn=206361-99-1 | |
| Description | CAS Common Chemistry is an open community resource for accessing chemical information. Nearly 500,000 chemical substances from CAS REGISTRY cover areas of community interest, including common and frequently regulated chemicals, and those relevant to high school and undergraduate chemistry classes. This chemical information, curated by our expert scientists, is provided in alignment with our mission as a division of the American Chemical Society. | |
| Explanation | The data from CAS Common Chemistry is provided under a CC-BY-NC 4.0 license, unless otherwise stated. | |
| Record name | Darunavir [USAN:INN:BAN] | |
| Source | ChemIDplus | |
| URL | https://pubchem.ncbi.nlm.nih.gov/substance/?source=chemidplus&sourceid=0206361991 | |
| Description | ChemIDplus is a free, web search system that provides access to the structure and nomenclature authority files used for the identification of chemical substances cited in National Library of Medicine (NLM) databases, including the TOXNET system. | |
| Record name | Darunavir | |
| Source | DrugBank | |
| URL | https://www.drugbank.ca/drugs/DB01264 | |
| Description | The DrugBank database is a unique bioinformatics and cheminformatics resource that combines detailed drug (i.e. chemical, pharmacological and pharmaceutical) data with comprehensive drug target (i.e. sequence, structure, and pathway) information. | |
| Explanation | Creative Common's Attribution-NonCommercial 4.0 International License (http://creativecommons.org/licenses/by-nc/4.0/legalcode) | |
| Record name | Darunavir | |
| Source | EPA DSSTox | |
| URL | https://comptox.epa.gov/dashboard/DTXSID0046779 | |
| Description | DSSTox provides a high quality public chemistry resource for supporting improved predictive toxicology. | |
| Record name | Carbamic acid, N-[(1S,2R)-3-[[(4-aminophenyl)sulfonyl](2-methylpropyl)amino]-2-hydroxy-1-(phenylmethyl)propyl]-, (3R,3aS,6aR)-hexahydrofuro[2,3-b]furan-3-yl ester | |
| Source | European Chemicals Agency (ECHA) | |
| URL | https://echa.europa.eu/information-on-chemicals | |
| Description | The European Chemicals Agency (ECHA) is an agency of the European Union which is the driving force among regulatory authorities in implementing the EU's groundbreaking chemicals legislation for the benefit of human health and the environment as well as for innovation and competitiveness. | |
| Explanation | Use of the information, documents and data from the ECHA website is subject to the terms and conditions of this Legal Notice, and subject to other binding limitations provided for under applicable law, the information, documents and data made available on the ECHA website may be reproduced, distributed and/or used, totally or in part, for non-commercial purposes provided that ECHA is acknowledged as the source: "Source: European Chemicals Agency, http://echa.europa.eu/". Such acknowledgement must be included in each copy of the material. ECHA permits and encourages organisations and individuals to create links to the ECHA website under the following cumulative conditions: Links can only be made to webpages that provide a link to the Legal Notice page. | |
| Record name | DARUNAVIR | |
| Source | FDA Global Substance Registration System (GSRS) | |
| URL | https://gsrs.ncats.nih.gov/ginas/app/beta/substances/YO603Y8113 | |
| Description | The FDA Global Substance Registration System (GSRS) enables the efficient and accurate exchange of information on what substances are in regulated products. Instead of relying on names, which vary across regulatory domains, countries, and regions, the GSRS knowledge base makes it possible for substances to be defined by standardized, scientific descriptions. | |
| Explanation | Unless otherwise noted, the contents of the FDA website (www.fda.gov), both text and graphics, are not copyrighted. They are in the public domain and may be republished, reprinted and otherwise used freely by anyone without the need to obtain permission from FDA. Credit to the U.S. Food and Drug Administration as the source is appreciated but not required. | |
| Record name | Darunavir | |
| Source | Hazardous Substances Data Bank (HSDB) | |
| URL | https://pubchem.ncbi.nlm.nih.gov/source/hsdb/7788 | |
| Description | The Hazardous Substances Data Bank (HSDB) is a toxicology database that focuses on the toxicology of potentially hazardous chemicals. It provides information on human exposure, industrial hygiene, emergency handling procedures, environmental fate, regulatory requirements, nanomaterials, and related areas. The information in HSDB has been assessed by a Scientific Review Panel. | |
| Record name | Darunavir | |
| Source | Human Metabolome Database (HMDB) | |
| URL | http://www.hmdb.ca/metabolites/HMDB0015393 | |
| Description | The Human Metabolome Database (HMDB) is a freely available electronic database containing detailed information about small molecule metabolites found in the human body. | |
| Explanation | HMDB is offered to the public as a freely available resource. Use and re-distribution of the data, in whole or in part, for commercial purposes requires explicit permission of the authors and explicit acknowledgment of the source material (HMDB) and the original publication (see the HMDB citing page). We ask that users who download significant portions of the database cite the HMDB paper in any resulting publications. | |
Melting Point |
74-76, 74 °C (decomposes) | |
| Record name | Darunavir | |
| Source | DrugBank | |
| URL | https://www.drugbank.ca/drugs/DB01264 | |
| Description | The DrugBank database is a unique bioinformatics and cheminformatics resource that combines detailed drug (i.e. chemical, pharmacological and pharmaceutical) data with comprehensive drug target (i.e. sequence, structure, and pathway) information. | |
| Explanation | Creative Common's Attribution-NonCommercial 4.0 International License (http://creativecommons.org/licenses/by-nc/4.0/legalcode) | |
| Record name | Darunavir | |
| Source | Hazardous Substances Data Bank (HSDB) | |
| URL | https://pubchem.ncbi.nlm.nih.gov/source/hsdb/7788 | |
| Description | The Hazardous Substances Data Bank (HSDB) is a toxicology database that focuses on the toxicology of potentially hazardous chemicals. It provides information on human exposure, industrial hygiene, emergency handling procedures, environmental fate, regulatory requirements, nanomaterials, and related areas. The information in HSDB has been assessed by a Scientific Review Panel. | |
Retrosynthesis Analysis
AI-Powered Synthesis Planning: Our tool employs the Template_relevance Pistachio, Template_relevance Bkms_metabolic, Template_relevance Pistachio_ringbreaker, Template_relevance Reaxys, Template_relevance Reaxys_biocatalysis model, leveraging a vast database of chemical reactions to predict feasible synthetic routes.
One-Step Synthesis Focus: Specifically designed for one-step synthesis, it provides concise and direct routes for your target compounds, streamlining the synthesis process.
Accurate Predictions: Utilizing the extensive PISTACHIO, BKMS_METABOLIC, PISTACHIO_RINGBREAKER, REAXYS, REAXYS_BIOCATALYSIS database, our tool offers high-accuracy predictions, reflecting the latest in chemical research and data.
Strategy Settings
| Precursor scoring | Relevance Heuristic |
|---|---|
| Min. plausibility | 0.01 |
| Model | Template_relevance |
| Template Set | Pistachio/Bkms_metabolic/Pistachio_ringbreaker/Reaxys/Reaxys_biocatalysis |
| Top-N result to add to graph | 6 |
Feasible Synthetic Routes
Haftungsausschluss und Informationen zu In-Vitro-Forschungsprodukten
Bitte beachten Sie, dass alle Artikel und Produktinformationen, die auf BenchChem präsentiert werden, ausschließlich zu Informationszwecken bestimmt sind. Die auf BenchChem zum Kauf angebotenen Produkte sind speziell für In-vitro-Studien konzipiert, die außerhalb lebender Organismen durchgeführt werden. In-vitro-Studien, abgeleitet von dem lateinischen Begriff "in Glas", beinhalten Experimente, die in kontrollierten Laborumgebungen unter Verwendung von Zellen oder Geweben durchgeführt werden. Es ist wichtig zu beachten, dass diese Produkte nicht als Arzneimittel oder Medikamente eingestuft sind und keine Zulassung der FDA für die Vorbeugung, Behandlung oder Heilung von medizinischen Zuständen, Beschwerden oder Krankheiten erhalten haben. Wir müssen betonen, dass jede Form der körperlichen Einführung dieser Produkte in Menschen oder Tiere gesetzlich strikt untersagt ist. Es ist unerlässlich, sich an diese Richtlinien zu halten, um die Einhaltung rechtlicher und ethischer Standards in Forschung und Experiment zu gewährleisten.