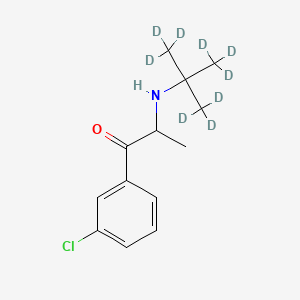
Bupropion D9
Descripción general
Descripción
Bupropion D9 is a deuterated form of bupropion, a norepinephrine and dopamine reuptake inhibitor commonly used in the treatment of major depressive disorder, seasonal affective disorder, and as an aid for smoking cessation . The deuterated form, this compound, is used primarily as an internal standard in analytical chemistry due to its stable isotopic labeling, which helps in the quantification of bupropion levels in biological matrices .
Mecanismo De Acción
Target of Action
Bupropion D9 primarily targets the norepinephrine transporter (NET) and the dopamine transporter (DAT) . These transporters are responsible for the reuptake of the neurotransmitters norepinephrine and dopamine from the synaptic cleft . This compound also acts as a non-competitive nicotine receptor antagonist .
Mode of Action
This compound exerts its pharmacological effects by weakly inhibiting the enzymes involved in the uptake of the neurotransmitters norepinephrine and dopamine from the synaptic cleft . This action prolongs the duration of these neurotransmitters within the neuronal synapse, leading to the downstream effects of these neurotransmitters . When used as an aid to smoking cessation, this compound is thought to confer its anti-craving and anti-withdrawal effects by inhibiting dopamine reuptake, which is thought to be involved in the reward pathways associated with nicotine .
Biochemical Pathways
This compound is hydroxylated to its primary active metabolite, hydroxybupropion, by the cytochrome P450 enzyme CYP2B6 . In vitro data suggest the existence of alternative hydroxylation pathways mediated by the highly polymorphic enzyme CYP2C19 . The majority of bupropion is cleared via reduction to threohydrobupropion (66%) and erythrohydrobupropion (6%) .
Pharmacokinetics
Following oral administration of bupropion hydrochloride tablets, peak plasma bupropion concentrations are usually achieved within 2 hours . Bupropion is extensively and rapidly absorbed in the gastrointestinal tract but experiences extensive first pass metabolism rendering its systemic bioavailability limited . Absorption is suggested to be between 80 and 90% .
Result of Action
The inhibition of norepinephrine and dopamine reuptake by this compound results in prolonged neurotransmitter action within the neuronal synapse, leading to increased locomotor activity and increased rates of responding in various schedule-controlled operant behavior tasks . When used as an aid to smoking cessation, this compound is thought to confer its anti-craving and anti-withdrawal effects by inhibiting dopamine reuptake .
Action Environment
The action, efficacy, and stability of this compound can be influenced by various environmental factors. It’s important to note that individual genetic variations, such as polymorphisms in metabolic enzymes, can significantly impact the pharmacokinetics and pharmacodynamics of this compound .
Análisis Bioquímico
Biochemical Properties
Bupropion D9 interacts with various enzymes and proteins within the body. It is primarily metabolized by the cytochrome P450 enzyme CYP2B6 into its primary active metabolite, hydroxybupropion . In vitro data suggest the existence of alternative hydroxylation pathways mediated by the highly polymorphic enzyme CYP2C19 .
Cellular Effects
This compound has been shown to have significant effects on various types of cells and cellular processes . It has been found to reduce negative biases in emotional processing but exacerbates impaired reward processing . It also has pro-inflammatory effects, particularly at doses of 50 μM and 100 μM .
Molecular Mechanism
This compound exerts its effects at the molecular level primarily by weakly inhibiting the enzymes involved in the uptake of the neurotransmitters norepinephrine and dopamine from the synaptic cleft . This prolongs their duration of action within the neuronal synapse and the downstream effects of these neurotransmitters .
Temporal Effects in Laboratory Settings
In laboratory settings, the effects of this compound have been observed to change over time . For instance, it was found that this compound initially further reduced the response bias for high-probability wins, but this effect reversed with six weeks’ treatment, normalizing reward processing .
Dosage Effects in Animal Models
The effects of this compound vary with different dosages in animal models . For instance, in isolation-induced aggression tests in mice, this compound increased time devoted to attack in long attack latency mice at doses of 10 mg/kg and 40 mg/kg .
Metabolic Pathways
This compound is involved in several metabolic pathways. It is primarily metabolized by the cytochrome P450 enzyme CYP2B6 into its primary active metabolite, hydroxybupropion . Cyp2c19 is also involved in this compound metabolism, primarily through alternate hydroxylation pathways .
Métodos De Preparación
Synthetic Routes and Reaction Conditions
The synthesis of Bupropion D9 involves the incorporation of deuterium atoms into the bupropion molecule. One common method is the hydrogen-deuterium exchange reaction, where hydrogen atoms in the bupropion molecule are replaced with deuterium atoms using deuterated reagents under specific conditions .
Industrial Production Methods
Industrial production of this compound typically involves large-scale hydrogen-deuterium exchange reactions. The process includes the use of deuterated solvents and catalysts to ensure the efficient incorporation of deuterium atoms. The reaction conditions are optimized to achieve high yields and purity of the deuterated product .
Análisis De Reacciones Químicas
Types of Reactions
Bupropion D9 undergoes similar chemical reactions as non-deuterated bupropion, including:
Oxidation: Bupropion can be oxidized to its metabolites, such as hydroxybupropion.
Reduction: Reduction reactions can convert bupropion to its reduced forms.
Substitution: Bupropion can undergo nucleophilic substitution reactions.
Common Reagents and Conditions
Oxidation: Common oxidizing agents include potassium permanganate and hydrogen peroxide.
Reduction: Reducing agents such as lithium aluminum hydride can be used.
Substitution: Nucleophilic reagents like sodium hydroxide are commonly used.
Major Products Formed
Oxidation: Hydroxybupropion and other oxidized metabolites.
Reduction: Reduced forms of bupropion.
Substitution: Various substituted derivatives depending on the nucleophile used
Aplicaciones Científicas De Investigación
Bupropion D9 is extensively used in scientific research, particularly in:
Analytical Chemistry: As an internal standard for the quantification of bupropion in biological samples using techniques like liquid chromatography-mass spectrometry (LC-MS) and gas chromatography-mass spectrometry (GC-MS)
Pharmacokinetics: Studying the pharmacokinetics and metabolism of bupropion in different formulations and release kinetics.
Clinical Research: Investigating the efficacy and safety of bupropion in various therapeutic applications, including depression and smoking cessation
Comparación Con Compuestos Similares
Similar Compounds
Vortioxetine: Another antidepressant that affects serotonin receptors and inhibits serotonin reuptake.
Cariprazine: An atypical antipsychotic used for schizophrenia and bipolar disorder.
Uniqueness
Bupropion D9 is unique due to its deuterated nature, which provides stability and precision in analytical measurements. Unlike other antidepressants, bupropion does not have significant serotonergic effects, making it less likely to cause sexual side effects, sedation, or weight gain . Its dual action on norepinephrine and dopamine reuptake, along with its nicotinic acetylcholine receptor antagonism, sets it apart from other antidepressants and smoking cessation aids .
Propiedades
IUPAC Name |
1-(3-chlorophenyl)-2-[[1,1,1,3,3,3-hexadeuterio-2-(trideuteriomethyl)propan-2-yl]amino]propan-1-one | |
|---|---|---|
| Details | Computed by Lexichem TK 2.7.0 (PubChem release 2021.05.07) | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
InChI |
InChI=1S/C13H18ClNO/c1-9(15-13(2,3)4)12(16)10-6-5-7-11(14)8-10/h5-9,15H,1-4H3/i2D3,3D3,4D3 | |
| Details | Computed by InChI 1.0.6 (PubChem release 2021.05.07) | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
InChI Key |
SNPPWIUOZRMYNY-WVZRYRIDSA-N | |
| Details | Computed by InChI 1.0.6 (PubChem release 2021.05.07) | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
Canonical SMILES |
CC(C(=O)C1=CC(=CC=C1)Cl)NC(C)(C)C | |
| Details | Computed by OEChem 2.3.0 (PubChem release 2021.05.07) | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
Isomeric SMILES |
[2H]C([2H])([2H])C(C([2H])([2H])[2H])(C([2H])([2H])[2H])NC(C)C(=O)C1=CC(=CC=C1)Cl | |
| Details | Computed by OEChem 2.3.0 (PubChem release 2021.05.07) | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
Molecular Formula |
C13H18ClNO | |
| Details | Computed by PubChem 2.1 (PubChem release 2021.05.07) | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
DSSTOX Substance ID |
DTXSID70649420 | |
| Record name | 1-(3-Chlorophenyl)-2-{[2-(~2~H_3_)methyl(~2~H_6_)propan-2-yl]amino}propan-1-one | |
| Source | EPA DSSTox | |
| URL | https://comptox.epa.gov/dashboard/DTXSID70649420 | |
| Description | DSSTox provides a high quality public chemistry resource for supporting improved predictive toxicology. | |
Molecular Weight |
248.79 g/mol | |
| Details | Computed by PubChem 2.1 (PubChem release 2021.05.07) | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
CAS No. |
150988-80-0 | |
| Record name | 1-(3-Chlorophenyl)-2-{[2-(~2~H_3_)methyl(~2~H_6_)propan-2-yl]amino}propan-1-one | |
| Source | EPA DSSTox | |
| URL | https://comptox.epa.gov/dashboard/DTXSID70649420 | |
| Description | DSSTox provides a high quality public chemistry resource for supporting improved predictive toxicology. | |
Synthesis routes and methods I
Procedure details




Synthesis routes and methods II
Procedure details




Synthesis routes and methods III
Procedure details













Synthesis routes and methods IV
Procedure details





Retrosynthesis Analysis
AI-Powered Synthesis Planning: Our tool employs the Template_relevance Pistachio, Template_relevance Bkms_metabolic, Template_relevance Pistachio_ringbreaker, Template_relevance Reaxys, Template_relevance Reaxys_biocatalysis model, leveraging a vast database of chemical reactions to predict feasible synthetic routes.
One-Step Synthesis Focus: Specifically designed for one-step synthesis, it provides concise and direct routes for your target compounds, streamlining the synthesis process.
Accurate Predictions: Utilizing the extensive PISTACHIO, BKMS_METABOLIC, PISTACHIO_RINGBREAKER, REAXYS, REAXYS_BIOCATALYSIS database, our tool offers high-accuracy predictions, reflecting the latest in chemical research and data.
Strategy Settings
| Precursor scoring | Relevance Heuristic |
|---|---|
| Min. plausibility | 0.01 |
| Model | Template_relevance |
| Template Set | Pistachio/Bkms_metabolic/Pistachio_ringbreaker/Reaxys/Reaxys_biocatalysis |
| Top-N result to add to graph | 6 |
Feasible Synthetic Routes
Descargo de responsabilidad e información sobre productos de investigación in vitro
Tenga en cuenta que todos los artículos e información de productos presentados en BenchChem están destinados únicamente con fines informativos. Los productos disponibles para la compra en BenchChem están diseñados específicamente para estudios in vitro, que se realizan fuera de organismos vivos. Los estudios in vitro, derivados del término latino "in vidrio", involucran experimentos realizados en entornos de laboratorio controlados utilizando células o tejidos. Es importante tener en cuenta que estos productos no se clasifican como medicamentos y no han recibido la aprobación de la FDA para la prevención, tratamiento o cura de ninguna condición médica, dolencia o enfermedad. Debemos enfatizar que cualquier forma de introducción corporal de estos productos en humanos o animales está estrictamente prohibida por ley. Es esencial adherirse a estas pautas para garantizar el cumplimiento de los estándares legales y éticos en la investigación y experimentación.
![4,4'-{Oxybis[benzene-4,1-diyl(hexafluoropropane-2,2-diyl)]}dianiline](/img/structure/B3031045.png)
![Benzyl 1,7-diazaspiro[3.5]nonane-1-carboxylate](/img/structure/B3031051.png)
![Methyl{[1-methyl-5-(trifluoromethyl)pyrazol-3-yl]methyl}amine](/img/structure/B3031055.png)