Propiltiouracilo
Descripción general
Descripción
Propiltiouracilo: es un agente antitiroideo de tiourea utilizado principalmente para tratar el hipertiroidismo, incluidas afecciones como la enfermedad de Graves y el bocio multinodular tóxico . Funciona inhibiendo la síntesis de las hormonas tiroideas y bloqueando la conversión de tiroxina (T4) a triyodotironina (T3) . El this compound se utiliza en medicina desde la década de 1940 y figura en la Lista de medicamentos esenciales de la Organización Mundial de la Salud .
Mecanismo De Acción
El propiltiouracilo ejerce sus efectos inhibiendo la enzima peroxidasa tiroidea, que es crucial para la yodación de los residuos de tirosina en la tiroglobulina, un paso clave en la síntesis de las hormonas tiroideas . Además, inhibe la conversión periférica de tiroxina (T4) a triyodotironina (T3), lo que reduce los niveles generales de hormonas tiroideas activas en el cuerpo .
Aplicaciones Científicas De Investigación
Química: El propiltiouracilo se utiliza como compuesto modelo en estudios que implican derivados de tiourea y sus propiedades químicas .
Biología: En la investigación biológica, el this compound se utiliza para estudiar la síntesis y el metabolismo de la hormona tiroidea .
Medicina: El this compound se utiliza ampliamente en el tratamiento del hipertiroidismo y la tirotoxicosis. También se utiliza en investigación para comprender los mecanismos de regulación de la hormona tiroidea .
Industria: En la industria farmacéutica, el this compound se utiliza en la formulación de medicamentos para trastornos de la tiroides .
Análisis Bioquímico
Biochemical Properties
Propylthiouracil plays a significant role in biochemical reactions. It inhibits the synthesis of thyroxine and blocks the peripheral conversion of thyroxine to tri-iodothyronine . This is achieved by binding to thyroid peroxidase, an enzyme that normally aids in the incorporation of iodine into the thyroglobulin protein to form thyroid hormones .
Cellular Effects
Propylthiouracil exerts various effects on cells and cellular processes. It decreases the amount of thyroid hormone produced by the thyroid gland, thereby influencing cell function . This includes impacts on cell signaling pathways, gene expression, and cellular metabolism .
Molecular Mechanism
The molecular mechanism of propylthiouracil involves its binding to thyroid peroxidase, thereby inhibiting the conversion of iodide to iodine . This action decreases thyroid hormone production and also interferes with the conversion of thyroxine (T4) to triiodothyronine (T3), reducing the activity of thyroid hormones .
Temporal Effects in Laboratory Settings
In laboratory settings, the effects of propylthiouracil change over time. For instance, a study showed that a 16-hour treatment with propylthiouracil was able to stimulate pregnenolone production in rat granulosa cells
Dosage Effects in Animal Models
The effects of propylthiouracil vary with different dosages in animal models. For instance, one study demonstrated that propylthiouracil, when supplemented with a low dose of levothyroxine, restored the propylthiouracil-induced reduction in the serum levels of T3 and T4 in rats .
Metabolic Pathways
Propylthiouracil is involved in the metabolic pathway of thyroid hormone production. It inhibits the synthesis of thyroxine and blocks the peripheral conversion of thyroxine to tri-iodothyronine .
Transport and Distribution
Propylthiouracil is readily absorbed and extensively metabolized. Approximately 35% of the drug is excreted in the urine, in intact and conjugated forms, within 24 hours .
Subcellular Localization
Given its mechanism of action, it is likely that propylthiouracil interacts with thyroid peroxidase, an enzyme located in the thyroid gland .
Métodos De Preparación
Rutas sintéticas y condiciones de reacción: El propiltiouracilo se puede sintetizar mediante la reacción de tiourea con yoduro de propilo en presencia de una base . La reacción normalmente implica calentar los reactivos bajo condiciones de reflujo para facilitar la formación del producto deseado.
Métodos de producción industrial: La producción industrial de this compound implica rutas sintéticas similares, pero a mayor escala. El proceso incluye la purificación del producto final mediante recristalización para garantizar una alta pureza y calidad .
Análisis De Reacciones Químicas
Tipos de reacciones:
Oxidación: El propiltiouracilo puede sufrir reacciones de oxidación, lo que lleva a la formación de enlaces disulfuro.
Reducción: Puede reducirse a su derivado tiol correspondiente.
Sustitución: El this compound puede participar en reacciones de sustitución, particularmente sustitución nucleófila.
Reactivos y condiciones comunes:
Oxidación: Peróxido de hidrógeno u otros agentes oxidantes.
Reducción: Agentes reductores como el borohidruro de sodio.
Sustitución: Nucleófilos como aminas o alcoholes.
Principales productos formados:
Oxidación: Derivados de disulfuro.
Reducción: Derivados de tiol.
Sustitución: Varios derivados de tiouracilo sustituidos.
Comparación Con Compuestos Similares
Compuestos similares:
Metimazol: Otro agente antitiroideo utilizado para tratar el hipertiroidismo.
Carbimazol: Un profármaco que se convierte en metimazol en el cuerpo.
Comparación:
Metimazol: A diferencia del propiltiouracilo, el metimazol no inhibe la conversión periférica de T4 a T3.
El this compound es único en su doble acción de inhibir tanto la síntesis de hormonas tiroideas como la conversión periférica, lo que lo hace particularmente útil en crisis tirotoxicas agudas .
Propiedades
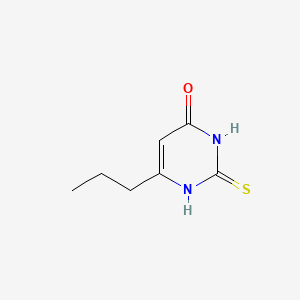
IUPAC Name |
6-propyl-2-sulfanylidene-1H-pyrimidin-4-one | |
|---|---|---|
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
InChI |
InChI=1S/C7H10N2OS/c1-2-3-5-4-6(10)9-7(11)8-5/h4H,2-3H2,1H3,(H2,8,9,10,11) | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
InChI Key |
KNAHARQHSZJURB-UHFFFAOYSA-N | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
Canonical SMILES |
CCCC1=CC(=O)NC(=S)N1 | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
Molecular Formula |
C7H10N2OS | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
DSSTOX Substance ID |
DTXSID5021209 | |
| Record name | 6-Propyl-2-thiouracil | |
| Source | EPA DSSTox | |
| URL | https://comptox.epa.gov/dashboard/DTXSID5021209 | |
| Description | DSSTox provides a high quality public chemistry resource for supporting improved predictive toxicology. | |
Molecular Weight |
170.23 g/mol | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
Physical Description |
Solid | |
| Record name | Propylthiouracil | |
| Source | Human Metabolome Database (HMDB) | |
| URL | http://www.hmdb.ca/metabolites/HMDB0014690 | |
| Description | The Human Metabolome Database (HMDB) is a freely available electronic database containing detailed information about small molecule metabolites found in the human body. | |
| Explanation | HMDB is offered to the public as a freely available resource. Use and re-distribution of the data, in whole or in part, for commercial purposes requires explicit permission of the authors and explicit acknowledgment of the source material (HMDB) and the original publication (see the HMDB citing page). We ask that users who download significant portions of the database cite the HMDB paper in any resulting publications. | |
Solubility |
>25.5 [ug/mL] (The mean of the results at pH 7.4), 1 part dissolves in about 900 parts water at 20 °C, in 100 parts boiling water, in 60 parts ethyl alc, in 60 parts acetone; practically insol in ether, chloroform, benzene; freely sol in aq soln of ammonia and alkali hydroxides, In water, 1204 mg/L at 25 °C, 4.66e-01 g/L | |
| Record name | SID855783 | |
| Source | Burnham Center for Chemical Genomics | |
| URL | https://pubchem.ncbi.nlm.nih.gov/bioassay/1996#section=Data-Table | |
| Description | Aqueous solubility in buffer at pH 7.4 | |
| Record name | Propylthiouracil | |
| Source | DrugBank | |
| URL | https://www.drugbank.ca/drugs/DB00550 | |
| Description | The DrugBank database is a unique bioinformatics and cheminformatics resource that combines detailed drug (i.e. chemical, pharmacological and pharmaceutical) data with comprehensive drug target (i.e. sequence, structure, and pathway) information. | |
| Explanation | Creative Common's Attribution-NonCommercial 4.0 International License (http://creativecommons.org/licenses/by-nc/4.0/legalcode) | |
| Record name | PROPYL THIOURACIL | |
| Source | Hazardous Substances Data Bank (HSDB) | |
| URL | https://pubchem.ncbi.nlm.nih.gov/source/hsdb/3390 | |
| Description | The Hazardous Substances Data Bank (HSDB) is a toxicology database that focuses on the toxicology of potentially hazardous chemicals. It provides information on human exposure, industrial hygiene, emergency handling procedures, environmental fate, regulatory requirements, nanomaterials, and related areas. The information in HSDB has been assessed by a Scientific Review Panel. | |
| Record name | Propylthiouracil | |
| Source | Human Metabolome Database (HMDB) | |
| URL | http://www.hmdb.ca/metabolites/HMDB0014690 | |
| Description | The Human Metabolome Database (HMDB) is a freely available electronic database containing detailed information about small molecule metabolites found in the human body. | |
| Explanation | HMDB is offered to the public as a freely available resource. Use and re-distribution of the data, in whole or in part, for commercial purposes requires explicit permission of the authors and explicit acknowledgment of the source material (HMDB) and the original publication (see the HMDB citing page). We ask that users who download significant portions of the database cite the HMDB paper in any resulting publications. | |
Mechanism of Action |
Propylthiouracil binds to thyroid peroxidase and thereby inhibits the conversion of iodide to iodine. Thyroid peroxidase normally converts iodide to iodine (via hydrogen peroxide as a cofactor) and also catalyzes the incorporation of the resulting iodide molecule onto both the 3 and/or 5 positions of the phenol rings of tyrosines found in thyroglobulin. Thyroglobulin is degraded to produce thyroxine (T4) and tri-iodothyronine (T3), which are the main hormones produced by the thyroid gland. Therefore propylthiouracil effectively inhibits the production of new thyroid hormones., Propylthiouracil inhibits the synthesis of thyroid hormones by interfering with the incorporation of iodine into tyrosyl residues of thyroglobulin; the drug also inhibits the coupling of these iodotyrosyl residues to form iodothyronine. Although the exact mechanism(s) has not been fully elucidated, propylthiouracil may interfere with the oxidation of iodide ion and iodotyrosyl groups. Based on limited evidence it appears that the coupling reaction is more sensitive to antithyroid agents than the iodination reaction. Propylthiouracil does not inhibit the action of thyroid hormones already formed and present in the thyroid gland or circulation nor does the drug interfere with the effectiveness of exogenously administered thyroid hormones. Patients whose thyroid gland contains relatively high concentration of iodine (e.g., from prior ingestion or from administration during diagnostic radiologic procedures) may respond relatively slowly to antithyroid agents. Unlike methimazole, propylthiouracil inhibits the peripheral deiodination of thyroxine to triiodothyronine. Although the importance of this inhibition has not been established, propylthiouracil has a theoretical advantage compared with methimazole or carbimazole in patients with thyrotoxic crisis, since a decreased rate of conversion of circulating thyroxine to triiodothyronine may be clinically beneficial in these patients., The thionamide /propylthiouracil/ inhibit organification of iodide and the coupling of iodotyrosines to form hormonally active iodothyronines., Inhibit synthesis of thyroid hormone within the thyroid gland by serving as substrates for thyroid peroxidase, which catalyzes the incorporation of oxidized iodide into tyrosine residues in thyroglobulin molecules and couples iodotyrosines. This diverts iodine from the synthesis of thyroid hormones. Antithyroid agents do not interfere with the actions of exogenous thyroid hormone or inhibit the release of thyroid hormones. Therefore, stores of thyroid hormones must be depleted before clinical effects will be apparent. Antithyroid agents may also have moderating effects on the underlying immunologic abnormalities, in hyperthyroidism due to Graves' disease (toxic-diffuse goiter), but evidence on this point reported to date is inconclusive., Type I 5'-deiodinase (D1) is inhibited by ... the antithyroid drug propylthiouracil., For more Mechanism of Action (Complete) data for PROPYL THIOURACIL (9 total), please visit the HSDB record page. | |
| Record name | Propylthiouracil | |
| Source | DrugBank | |
| URL | https://www.drugbank.ca/drugs/DB00550 | |
| Description | The DrugBank database is a unique bioinformatics and cheminformatics resource that combines detailed drug (i.e. chemical, pharmacological and pharmaceutical) data with comprehensive drug target (i.e. sequence, structure, and pathway) information. | |
| Explanation | Creative Common's Attribution-NonCommercial 4.0 International License (http://creativecommons.org/licenses/by-nc/4.0/legalcode) | |
| Record name | PROPYL THIOURACIL | |
| Source | Hazardous Substances Data Bank (HSDB) | |
| URL | https://pubchem.ncbi.nlm.nih.gov/source/hsdb/3390 | |
| Description | The Hazardous Substances Data Bank (HSDB) is a toxicology database that focuses on the toxicology of potentially hazardous chemicals. It provides information on human exposure, industrial hygiene, emergency handling procedures, environmental fate, regulatory requirements, nanomaterials, and related areas. The information in HSDB has been assessed by a Scientific Review Panel. | |
Impurities |
... Small amounts of thiourea present as an impurity., Small amounts of thiourea may be present in propylthiouracil as an impurity. | |
| Record name | PROPYL THIOURACIL | |
| Source | Hazardous Substances Data Bank (HSDB) | |
| URL | https://pubchem.ncbi.nlm.nih.gov/source/hsdb/3390 | |
| Description | The Hazardous Substances Data Bank (HSDB) is a toxicology database that focuses on the toxicology of potentially hazardous chemicals. It provides information on human exposure, industrial hygiene, emergency handling procedures, environmental fate, regulatory requirements, nanomaterials, and related areas. The information in HSDB has been assessed by a Scientific Review Panel. | |
Color/Form |
White crystalline powder of starch-like appearance to eye and to touch | |
CAS No. |
51-52-5 | |
| Record name | Propylthiouracil | |
| Source | CAS Common Chemistry | |
| URL | https://commonchemistry.cas.org/detail?cas_rn=51-52-5 | |
| Description | CAS Common Chemistry is an open community resource for accessing chemical information. Nearly 500,000 chemical substances from CAS REGISTRY cover areas of community interest, including common and frequently regulated chemicals, and those relevant to high school and undergraduate chemistry classes. This chemical information, curated by our expert scientists, is provided in alignment with our mission as a division of the American Chemical Society. | |
| Explanation | The data from CAS Common Chemistry is provided under a CC-BY-NC 4.0 license, unless otherwise stated. | |
| Record name | Propylthiouracil [USP:INN:BAN:JAN] | |
| Source | ChemIDplus | |
| URL | https://pubchem.ncbi.nlm.nih.gov/substance/?source=chemidplus&sourceid=0000051525 | |
| Description | ChemIDplus is a free, web search system that provides access to the structure and nomenclature authority files used for the identification of chemical substances cited in National Library of Medicine (NLM) databases, including the TOXNET system. | |
| Record name | Propylthiouracil | |
| Source | DrugBank | |
| URL | https://www.drugbank.ca/drugs/DB00550 | |
| Description | The DrugBank database is a unique bioinformatics and cheminformatics resource that combines detailed drug (i.e. chemical, pharmacological and pharmaceutical) data with comprehensive drug target (i.e. sequence, structure, and pathway) information. | |
| Explanation | Creative Common's Attribution-NonCommercial 4.0 International License (http://creativecommons.org/licenses/by-nc/4.0/legalcode) | |
| Record name | propylthiouracil | |
| Source | DTP/NCI | |
| URL | https://dtp.cancer.gov/dtpstandard/servlet/dwindex?searchtype=NSC&outputformat=html&searchlist=757302 | |
| Description | The NCI Development Therapeutics Program (DTP) provides services and resources to the academic and private-sector research communities worldwide to facilitate the discovery and development of new cancer therapeutic agents. | |
| Explanation | Unless otherwise indicated, all text within NCI products is free of copyright and may be reused without our permission. Credit the National Cancer Institute as the source. | |
| Record name | propylthiouracil | |
| Source | DTP/NCI | |
| URL | https://dtp.cancer.gov/dtpstandard/servlet/dwindex?searchtype=NSC&outputformat=html&searchlist=70461 | |
| Description | The NCI Development Therapeutics Program (DTP) provides services and resources to the academic and private-sector research communities worldwide to facilitate the discovery and development of new cancer therapeutic agents. | |
| Explanation | Unless otherwise indicated, all text within NCI products is free of copyright and may be reused without our permission. Credit the National Cancer Institute as the source. | |
| Record name | propylthiouracil | |
| Source | DTP/NCI | |
| URL | https://dtp.cancer.gov/dtpstandard/servlet/dwindex?searchtype=NSC&outputformat=html&searchlist=6498 | |
| Description | The NCI Development Therapeutics Program (DTP) provides services and resources to the academic and private-sector research communities worldwide to facilitate the discovery and development of new cancer therapeutic agents. | |
| Explanation | Unless otherwise indicated, all text within NCI products is free of copyright and may be reused without our permission. Credit the National Cancer Institute as the source. | |
| Record name | 6-Propyl-2-thiouracil | |
| Source | EPA DSSTox | |
| URL | https://comptox.epa.gov/dashboard/DTXSID5021209 | |
| Description | DSSTox provides a high quality public chemistry resource for supporting improved predictive toxicology. | |
| Record name | Propylthiouracil | |
| Source | European Chemicals Agency (ECHA) | |
| URL | https://echa.europa.eu/substance-information/-/substanceinfo/100.000.095 | |
| Description | The European Chemicals Agency (ECHA) is an agency of the European Union which is the driving force among regulatory authorities in implementing the EU's groundbreaking chemicals legislation for the benefit of human health and the environment as well as for innovation and competitiveness. | |
| Explanation | Use of the information, documents and data from the ECHA website is subject to the terms and conditions of this Legal Notice, and subject to other binding limitations provided for under applicable law, the information, documents and data made available on the ECHA website may be reproduced, distributed and/or used, totally or in part, for non-commercial purposes provided that ECHA is acknowledged as the source: "Source: European Chemicals Agency, http://echa.europa.eu/". Such acknowledgement must be included in each copy of the material. ECHA permits and encourages organisations and individuals to create links to the ECHA website under the following cumulative conditions: Links can only be made to webpages that provide a link to the Legal Notice page. | |
| Record name | PROPYLTHIOURACIL | |
| Source | FDA Global Substance Registration System (GSRS) | |
| URL | https://gsrs.ncats.nih.gov/ginas/app/beta/substances/721M9407IY | |
| Description | The FDA Global Substance Registration System (GSRS) enables the efficient and accurate exchange of information on what substances are in regulated products. Instead of relying on names, which vary across regulatory domains, countries, and regions, the GSRS knowledge base makes it possible for substances to be defined by standardized, scientific descriptions. | |
| Explanation | Unless otherwise noted, the contents of the FDA website (www.fda.gov), both text and graphics, are not copyrighted. They are in the public domain and may be republished, reprinted and otherwise used freely by anyone without the need to obtain permission from FDA. Credit to the U.S. Food and Drug Administration as the source is appreciated but not required. | |
| Record name | PROPYL THIOURACIL | |
| Source | Hazardous Substances Data Bank (HSDB) | |
| URL | https://pubchem.ncbi.nlm.nih.gov/source/hsdb/3390 | |
| Description | The Hazardous Substances Data Bank (HSDB) is a toxicology database that focuses on the toxicology of potentially hazardous chemicals. It provides information on human exposure, industrial hygiene, emergency handling procedures, environmental fate, regulatory requirements, nanomaterials, and related areas. The information in HSDB has been assessed by a Scientific Review Panel. | |
| Record name | Propylthiouracil | |
| Source | Human Metabolome Database (HMDB) | |
| URL | http://www.hmdb.ca/metabolites/HMDB0014690 | |
| Description | The Human Metabolome Database (HMDB) is a freely available electronic database containing detailed information about small molecule metabolites found in the human body. | |
| Explanation | HMDB is offered to the public as a freely available resource. Use and re-distribution of the data, in whole or in part, for commercial purposes requires explicit permission of the authors and explicit acknowledgment of the source material (HMDB) and the original publication (see the HMDB citing page). We ask that users who download significant portions of the database cite the HMDB paper in any resulting publications. | |
Melting Point |
219-221 °C, 219 °C | |
| Record name | Propylthiouracil | |
| Source | DrugBank | |
| URL | https://www.drugbank.ca/drugs/DB00550 | |
| Description | The DrugBank database is a unique bioinformatics and cheminformatics resource that combines detailed drug (i.e. chemical, pharmacological and pharmaceutical) data with comprehensive drug target (i.e. sequence, structure, and pathway) information. | |
| Explanation | Creative Common's Attribution-NonCommercial 4.0 International License (http://creativecommons.org/licenses/by-nc/4.0/legalcode) | |
| Record name | PROPYL THIOURACIL | |
| Source | Hazardous Substances Data Bank (HSDB) | |
| URL | https://pubchem.ncbi.nlm.nih.gov/source/hsdb/3390 | |
| Description | The Hazardous Substances Data Bank (HSDB) is a toxicology database that focuses on the toxicology of potentially hazardous chemicals. It provides information on human exposure, industrial hygiene, emergency handling procedures, environmental fate, regulatory requirements, nanomaterials, and related areas. The information in HSDB has been assessed by a Scientific Review Panel. | |
| Record name | Propylthiouracil | |
| Source | Human Metabolome Database (HMDB) | |
| URL | http://www.hmdb.ca/metabolites/HMDB0014690 | |
| Description | The Human Metabolome Database (HMDB) is a freely available electronic database containing detailed information about small molecule metabolites found in the human body. | |
| Explanation | HMDB is offered to the public as a freely available resource. Use and re-distribution of the data, in whole or in part, for commercial purposes requires explicit permission of the authors and explicit acknowledgment of the source material (HMDB) and the original publication (see the HMDB citing page). We ask that users who download significant portions of the database cite the HMDB paper in any resulting publications. | |
Retrosynthesis Analysis
AI-Powered Synthesis Planning: Our tool employs the Template_relevance Pistachio, Template_relevance Bkms_metabolic, Template_relevance Pistachio_ringbreaker, Template_relevance Reaxys, Template_relevance Reaxys_biocatalysis model, leveraging a vast database of chemical reactions to predict feasible synthetic routes.
One-Step Synthesis Focus: Specifically designed for one-step synthesis, it provides concise and direct routes for your target compounds, streamlining the synthesis process.
Accurate Predictions: Utilizing the extensive PISTACHIO, BKMS_METABOLIC, PISTACHIO_RINGBREAKER, REAXYS, REAXYS_BIOCATALYSIS database, our tool offers high-accuracy predictions, reflecting the latest in chemical research and data.
Strategy Settings
| Precursor scoring | Relevance Heuristic |
|---|---|
| Min. plausibility | 0.01 |
| Model | Template_relevance |
| Template Set | Pistachio/Bkms_metabolic/Pistachio_ringbreaker/Reaxys/Reaxys_biocatalysis |
| Top-N result to add to graph | 6 |
Feasible Synthetic Routes
A: Propylthiouracil inhibits the enzyme thyroid peroxidase, which is essential for thyroid hormone synthesis. [, , ] This enzyme catalyzes the oxidation of iodide and its incorporation into tyrosine residues within thyroglobulin, ultimately leading to the formation of thyroxine (T4) and triiodothyronine (T3). By blocking thyroid peroxidase, Propylthiouracil reduces the production of T4 and T3. [] Additionally, Propylthiouracil specifically hinders the conversion of T4 to T3 in peripheral tissues. []
A: While Propylthiouracil primarily inhibits thyroid hormone synthesis, studies in amphibian thyroid gland explant cultures demonstrate that it also effectively inhibits thyroxine (T4) release. [] This inhibition was dose-dependent and observed alongside the inhibitory effects of other known thyroid disruptors like methimazole and perchlorate. []
ANone: Propylthiouracil has a molecular formula of C7H10N2OS and a molecular weight of 170.24 g/mol.
A: While specific stability data is limited in the provided research, a high-performance liquid chromatography method developed for Propylthiouracil determination highlights its stability under extreme acidic and basic conditions. [] This suggests the method's specificity for Propylthiouracil and its potential for use in stability studies. []
A: Research indicates that hyperthyroidism accelerates the metabolism of Propylthiouracil, leading to a shortened plasma half-life. [] Conversely, in hypothyroidism, Propylthiouracil metabolism is slowed, resulting in a prolonged plasma half-life. [] These alterations appear primarily due to changes in hepatic microsomal enzyme activity influenced by thyroid hormone levels. []
A: A study on children with hyperthyroidism demonstrated that topical injection of dexamethasone combined with oral Propylthiouracil is more effective than oral Propylthiouracil alone in achieving symptom remission and normalizing thyroid hormone levels. []
ANone: While Propylthiouracil is generally considered safe for treating hyperthyroidism, it has been associated with several adverse effects, including:
- Hepatotoxicity: Cases of Propylthiouracil-induced liver injury, ranging from mild hepatitis to acute liver failure, have been reported. [, , ]
- Agranulocytosis: This rare but serious side effect involves a severe decrease in white blood cells, increasing the risk of infections. [, , , ]
- Vasculitis: Propylthiouracil has been linked to vasculitis, an inflammation of blood vessels, potentially leading to organ damage. [, , , , , ]
- Antineutrophil cytoplasmic antibody (ANCA)-associated vasculitis: Propylthiouracil has been associated with ANCA-positive vasculitis, which can manifest with various symptoms depending on the affected organs. [, , , , ]
A: High-performance liquid chromatography (HPLC) has been used to develop a rapid and sensitive method for determining Propylthiouracil concentration. [] This method exhibits good linearity, precision, accuracy, and stability-indicating properties, making it suitable for pharmaceutical analysis. []
A: Propylthiouracil has been shown to inhibit hepatic microsomal drug metabolism, as evidenced by the prolonged plasma half-lives of drugs like antipyrine and methimazole in hypothyroid patients treated with Propylthiouracil. [] This effect is likely related to Propylthiouracil's influence on thyroid hormone levels, as returning to a euthyroid state restores drug metabolism towards normal. []
A: Methimazole and carbimazole are other thionamide antithyroid drugs commonly used as alternatives to Propylthiouracil. [, ] While they share a similar mechanism of action, their pharmacokinetic profiles and potential side effects differ, prompting careful consideration when choosing the appropriate treatment option. [, ]
A: Research is ongoing to develop new therapeutic options for hyperthyroidism. One promising avenue is the development of TSH receptor antagonists, such as Compound 52. [] This low-molecular-weight antagonist selectively inhibits TSH receptor activation, potentially offering a more targeted approach with fewer side effects compared to traditional antithyroid drugs. []
A: Antithyroid drugs, including Propylthiouracil, have been used to treat hyperthyroidism for over 65 years. [] Their introduction marked a significant milestone in managing hyperthyroidism, offering a medical alternative to surgical removal or radioiodine ablation of the thyroid gland. []
Descargo de responsabilidad e información sobre productos de investigación in vitro
Tenga en cuenta que todos los artículos e información de productos presentados en BenchChem están destinados únicamente con fines informativos. Los productos disponibles para la compra en BenchChem están diseñados específicamente para estudios in vitro, que se realizan fuera de organismos vivos. Los estudios in vitro, derivados del término latino "in vidrio", involucran experimentos realizados en entornos de laboratorio controlados utilizando células o tejidos. Es importante tener en cuenta que estos productos no se clasifican como medicamentos y no han recibido la aprobación de la FDA para la prevención, tratamiento o cura de ninguna condición médica, dolencia o enfermedad. Debemos enfatizar que cualquier forma de introducción corporal de estos productos en humanos o animales está estrictamente prohibida por ley. Es esencial adherirse a estas pautas para garantizar el cumplimiento de los estándares legales y éticos en la investigación y experimentación.