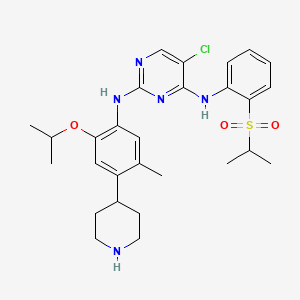
Ertugliflozina
Descripción general
Descripción
La ertugliflozina es un inhibidor selectivo del cotransportador 2 de sodio-glucosa (SGLT2), utilizado principalmente para el tratamiento de la diabetes mellitus tipo 2. Funciona bloqueando la reabsorción de glucosa en los riñones, lo que lleva a una mayor excreción de glucosa a través de la orina. Esto ayuda a reducir los niveles de glucosa en sangre en pacientes con diabetes tipo 2 .
Aplicaciones Científicas De Investigación
La ertugliflozina tiene una amplia gama de aplicaciones de investigación científica, particularmente en los campos de la química, la biología, la medicina y la industria. En medicina, se utiliza para controlar la diabetes mellitus tipo 2 mejorando el control glucémico. También se ha estudiado por sus posibles beneficios cardiovasculares, incluyendo la reducción del riesgo de eventos cardiovasculares adversos importantes y las hospitalizaciones por insuficiencia cardíaca . En química, se utiliza como un compuesto modelo para estudiar la farmacocinética y la farmacodinamia de los inhibidores de SGLT2 . En la industria, se utiliza en el desarrollo de nuevos medicamentos y formulaciones antidiabéticos .
Mecanismo De Acción
La ertugliflozina ejerce sus efectos inhibiendo el cotransportador 2 de sodio-glucosa (SGLT2) en los riñones. Esta inhibición previene la reabsorción de glucosa del filtrado glomerular, lo que lleva a una mayor excreción de glucosa urinaria y una reducción de los niveles de glucosa en sangre . Los objetivos moleculares implicados en este proceso incluyen las proteínas SGLT2 ubicadas en los túbulos proximales de los riñones .
Análisis Bioquímico
Biochemical Properties
Ertugliflozin plays a crucial role in biochemical reactions by inhibiting the sodium-glucose cotransporter 2, which is predominantly expressed in the proximal renal tubules. This inhibition prevents the reabsorption of glucose from the glomerular filtrate back into the bloodstream, thereby increasing urinary glucose excretion . Ertugliflozin interacts with several biomolecules, including the sodium-glucose cotransporter 2 protein itself. The binding of ertugliflozin to sodium-glucose cotransporter 2 is highly selective, ensuring minimal interaction with other glucose transporters .
Cellular Effects
Ertugliflozin exerts significant effects on various cell types, particularly renal tubular cells. By inhibiting sodium-glucose cotransporter 2, ertugliflozin reduces glucose reabsorption, leading to increased glucose excretion and decreased blood glucose levels . This reduction in glucose levels can influence cell signaling pathways, such as the insulin signaling pathway, and can lead to changes in gene expression related to glucose metabolism . Additionally, ertugliflozin has been shown to reduce oxidative stress and inflammation in renal cells, contributing to its protective effects on kidney function .
Molecular Mechanism
The molecular mechanism of action of ertugliflozin involves the selective inhibition of sodium-glucose cotransporter 2. Ertugliflozin binds to the sodium-glucose cotransporter 2 protein, blocking its ability to transport glucose and sodium ions across the renal tubular cell membrane . This inhibition leads to increased glucose excretion in the urine and decreased blood glucose levels. Ertugliflozin also affects the expression of genes involved in glucose metabolism and insulin signaling, further contributing to its hypoglycemic effects .
Temporal Effects in Laboratory Settings
In laboratory settings, the effects of ertugliflozin have been observed to change over time. Ertugliflozin is rapidly absorbed following oral administration, with peak plasma concentrations occurring within 1 to 2 hours . The terminal elimination half-life of ertugliflozin ranges from 11 to 18 hours, allowing for once-daily dosing . Long-term studies have shown that ertugliflozin maintains its efficacy in reducing blood glucose levels over extended periods, with minimal degradation or loss of stability . Additionally, ertugliflozin has been shown to have long-term protective effects on renal function in both in vitro and in vivo studies .
Dosage Effects in Animal Models
In animal models, the effects of ertugliflozin vary with different dosages. Studies have shown that lower doses of ertugliflozin effectively reduce blood glucose levels without causing significant adverse effects . Higher doses of ertugliflozin can lead to increased urinary glucose excretion and potential adverse effects, such as dehydration and electrolyte imbalances . Threshold effects have been observed, with a plateau in glucose-lowering effects at higher doses . Toxicity studies in animal models have shown that ertugliflozin is generally well-tolerated, with a low incidence of adverse effects at therapeutic doses .
Metabolic Pathways
Ertugliflozin is primarily metabolized through glucuronidation, mediated by the enzymes uridine 5’-diphospho-glucuronosyltransferase 1A9 and uridine 5’-diphospho-glucuronosyltransferase 2B7 . Approximately 86% of ertugliflozin is metabolized via this pathway, resulting in the formation of pharmacologically inactive glucuronide conjugates . A minor portion of ertugliflozin undergoes oxidative metabolism by cytochrome P450 3A4, contributing to about 12% of its clearance . The metabolic pathways of ertugliflozin ensure its efficient elimination from the body, with minimal accumulation of active drug .
Transport and Distribution
Ertugliflozin is rapidly absorbed following oral administration, with peak plasma concentrations occurring within 1 to 2 hours . The drug is widely distributed throughout the body, with a volume of distribution of approximately 85 liters . Ertugliflozin is highly bound to plasma proteins, primarily albumin, which facilitates its transport and distribution within the bloodstream . The primary route of elimination for ertugliflozin is through metabolism, with minimal renal excretion of the unchanged drug .
Subcellular Localization
Ertugliflozin primarily localizes to the renal proximal tubular cells, where it exerts its inhibitory effects on sodium-glucose cotransporter 2 . The drug’s localization to these cells is facilitated by its high affinity for the sodium-glucose cotransporter 2 protein, ensuring targeted inhibition of glucose reabsorption . Ertugliflozin does not undergo significant post-translational modifications or targeting signals that direct it to other cellular compartments or organelles .
Métodos De Preparación
Rutas sintéticas y condiciones de reacción: La síntesis de ertugliflozina implica varios pasos, comenzando con la protección del grupo alcohol primario de un compuesto intermedio con un grupo tritilo en presencia de piridina. Esto va seguido de una posterior desprotección con ácido p-toluensulfónico . El proceso también implica el uso de compuestos de Fórmula III, Fórmula IV y Fórmula VII, que están protegidos con grupos protectores adecuados para garantizar una alta pureza y rendimiento .
Métodos de producción industrial: La producción industrial de this compound sigue rutas sintéticas similares pero a mayor escala. El proceso implica el uso de técnicas y equipos avanzados para garantizar un alto rendimiento y pureza. El uso de productos químicos peligrosos como la piridina se minimiza para garantizar la seguridad y el cumplimiento de las normas industriales .
Análisis De Reacciones Químicas
Tipos de reacciones: La ertugliflozina experimenta diversas reacciones químicas, incluyendo oxidación, reducción y sustitución. Es estable en condiciones ácidas, alcalinas y térmicas, pero muestra una degradación notable en condiciones fotóliticas y oxidativas .
Reactivos y condiciones comunes: Los reactivos comunes utilizados en las reacciones que involucran this compound incluyen ácido p-toluensulfónico para la desprotección y ácido ortofosfórico para el ajuste del pH. Las reacciones se llevan a cabo normalmente en condiciones controladas para asegurar la estabilidad y pureza del compuesto .
Principales productos formados: Los principales productos formados a partir de las reacciones que involucran this compound incluyen sus metabolitos, que se excretan principalmente a través de la orina. La principal vía metabólica es la glucuronidación, que representa aproximadamente el 86% del metabolismo .
Comparación Con Compuestos Similares
La ertugliflozina forma parte de la clase de medicamentos inhibidores de SGLT2, que también incluye canagliflozina, dapagliflozina y empagliflozina. En comparación con estos compuestos, la this compound ha mostrado una eficacia similar en la reducción de los niveles de glucosa en sangre, pero puede tener un perfil de seguridad y efectos secundarios diferente . Por ejemplo, se ha encontrado que la this compound tiene un menor riesgo de causar infecciones del tracto urinario en comparación con algunos otros inhibidores de SGLT2 .
Lista de compuestos similares:- Canagliflozina
- Dapagliflozina
- Empagliflozina
- Bexagliflozina
Propiedades
IUPAC Name |
(1S,2S,3S,4R,5S)-5-[4-chloro-3-[(4-ethoxyphenyl)methyl]phenyl]-1-(hydroxymethyl)-6,8-dioxabicyclo[3.2.1]octane-2,3,4-triol | |
|---|---|---|
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
InChI |
InChI=1S/C22H25ClO7/c1-2-28-16-6-3-13(4-7-16)9-14-10-15(5-8-17(14)23)22-20(27)18(25)19(26)21(11-24,30-22)12-29-22/h3-8,10,18-20,24-27H,2,9,11-12H2,1H3/t18-,19-,20+,21-,22-/m0/s1 | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
InChI Key |
MCIACXAZCBVDEE-CUUWFGFTSA-N | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
Canonical SMILES |
CCOC1=CC=C(C=C1)CC2=C(C=CC(=C2)C34C(C(C(C(O3)(CO4)CO)O)O)O)Cl | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
Isomeric SMILES |
CCOC1=CC=C(C=C1)CC2=C(C=CC(=C2)[C@@]34[C@@H]([C@H]([C@@H]([C@@](O3)(CO4)CO)O)O)O)Cl | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
Molecular Formula |
C22H25ClO7 | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
DSSTOX Substance ID |
DTXSID40153120 | |
| Record name | PF-04971729 | |
| Source | EPA DSSTox | |
| URL | https://comptox.epa.gov/dashboard/DTXSID40153120 | |
| Description | DSSTox provides a high quality public chemistry resource for supporting improved predictive toxicology. | |
Molecular Weight |
436.9 g/mol | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
Solubility |
Very slightly soluble | |
| Record name | Ertugliflozin | |
| Source | DrugBank | |
| URL | https://www.drugbank.ca/drugs/DB11827 | |
| Description | The DrugBank database is a unique bioinformatics and cheminformatics resource that combines detailed drug (i.e. chemical, pharmacological and pharmaceutical) data with comprehensive drug target (i.e. sequence, structure, and pathway) information. | |
| Explanation | Creative Common's Attribution-NonCommercial 4.0 International License (http://creativecommons.org/licenses/by-nc/4.0/legalcode) | |
Mechanism of Action |
As part of a normal process, the glucose from the blood is filtered for excretion and reabsorbed in the glomerulus so less than one percent of this glucose is excreted in the urine. The reabsorption is mediated by the sodium-dependent glucose cotransporter (SGLT), mainly the type 2 which is responsible for 90% of the reabsorbed glucose. Ertugliflozin is a small inhibitor of the SGLT2 and its activity increases glucose excretion, reducing hyperglycemia without the requirement of excessive insulin secretion. | |
| Record name | Ertugliflozin | |
| Source | DrugBank | |
| URL | https://www.drugbank.ca/drugs/DB11827 | |
| Description | The DrugBank database is a unique bioinformatics and cheminformatics resource that combines detailed drug (i.e. chemical, pharmacological and pharmaceutical) data with comprehensive drug target (i.e. sequence, structure, and pathway) information. | |
| Explanation | Creative Common's Attribution-NonCommercial 4.0 International License (http://creativecommons.org/licenses/by-nc/4.0/legalcode) | |
CAS No. |
1210344-57-2, 1431329-06-4, 1210344-83-4 | |
| Record name | Ertugliflozin | |
| Source | CAS Common Chemistry | |
| URL | https://commonchemistry.cas.org/detail?cas_rn=1210344-57-2 | |
| Description | CAS Common Chemistry is an open community resource for accessing chemical information. Nearly 500,000 chemical substances from CAS REGISTRY cover areas of community interest, including common and frequently regulated chemicals, and those relevant to high school and undergraduate chemistry classes. This chemical information, curated by our expert scientists, is provided in alignment with our mission as a division of the American Chemical Society. | |
| Explanation | The data from CAS Common Chemistry is provided under a CC-BY-NC 4.0 license, unless otherwise stated. | |
| Record name | Ertugliflozin [USAN:INN] | |
| Source | ChemIDplus | |
| URL | https://pubchem.ncbi.nlm.nih.gov/substance/?source=chemidplus&sourceid=1210344572 | |
| Description | ChemIDplus is a free, web search system that provides access to the structure and nomenclature authority files used for the identification of chemical substances cited in National Library of Medicine (NLM) databases, including the TOXNET system. | |
| Record name | Ertugliflozin | |
| Source | DrugBank | |
| URL | https://www.drugbank.ca/drugs/DB11827 | |
| Description | The DrugBank database is a unique bioinformatics and cheminformatics resource that combines detailed drug (i.e. chemical, pharmacological and pharmaceutical) data with comprehensive drug target (i.e. sequence, structure, and pathway) information. | |
| Explanation | Creative Common's Attribution-NonCommercial 4.0 International License (http://creativecommons.org/licenses/by-nc/4.0/legalcode) | |
| Record name | PF-04971729 | |
| Source | EPA DSSTox | |
| URL | https://comptox.epa.gov/dashboard/DTXSID40153120 | |
| Description | DSSTox provides a high quality public chemistry resource for supporting improved predictive toxicology. | |
| Record name | 1,6-Anhydro-1-C-[4-chloro-3-[(4-ethoxyphenyl)methyl]phenyl]-5-C-(hydroxymethyl)-beta-L-idopyranose | |
| Source | European Chemicals Agency (ECHA) | |
| URL | https://echa.europa.eu/information-on-chemicals | |
| Description | The European Chemicals Agency (ECHA) is an agency of the European Union which is the driving force among regulatory authorities in implementing the EU's groundbreaking chemicals legislation for the benefit of human health and the environment as well as for innovation and competitiveness. | |
| Explanation | Use of the information, documents and data from the ECHA website is subject to the terms and conditions of this Legal Notice, and subject to other binding limitations provided for under applicable law, the information, documents and data made available on the ECHA website may be reproduced, distributed and/or used, totally or in part, for non-commercial purposes provided that ECHA is acknowledged as the source: "Source: European Chemicals Agency, http://echa.europa.eu/". Such acknowledgement must be included in each copy of the material. ECHA permits and encourages organisations and individuals to create links to the ECHA website under the following cumulative conditions: Links can only be made to webpages that provide a link to the Legal Notice page. | |
| Record name | Ertugliflozin | |
| Source | European Chemicals Agency (ECHA) | |
| URL | https://echa.europa.eu/information-on-chemicals | |
| Description | The European Chemicals Agency (ECHA) is an agency of the European Union which is the driving force among regulatory authorities in implementing the EU's groundbreaking chemicals legislation for the benefit of human health and the environment as well as for innovation and competitiveness. | |
| Explanation | Use of the information, documents and data from the ECHA website is subject to the terms and conditions of this Legal Notice, and subject to other binding limitations provided for under applicable law, the information, documents and data made available on the ECHA website may be reproduced, distributed and/or used, totally or in part, for non-commercial purposes provided that ECHA is acknowledged as the source: "Source: European Chemicals Agency, http://echa.europa.eu/". Such acknowledgement must be included in each copy of the material. ECHA permits and encourages organisations and individuals to create links to the ECHA website under the following cumulative conditions: Links can only be made to webpages that provide a link to the Legal Notice page. | |
| Record name | Ertugliflozin | |
| Source | European Chemicals Agency (ECHA) | |
| URL | https://echa.europa.eu/information-on-chemicals | |
| Description | The European Chemicals Agency (ECHA) is an agency of the European Union which is the driving force among regulatory authorities in implementing the EU's groundbreaking chemicals legislation for the benefit of human health and the environment as well as for innovation and competitiveness. | |
| Explanation | Use of the information, documents and data from the ECHA website is subject to the terms and conditions of this Legal Notice, and subject to other binding limitations provided for under applicable law, the information, documents and data made available on the ECHA website may be reproduced, distributed and/or used, totally or in part, for non-commercial purposes provided that ECHA is acknowledged as the source: "Source: European Chemicals Agency, http://echa.europa.eu/". Such acknowledgement must be included in each copy of the material. ECHA permits and encourages organisations and individuals to create links to the ECHA website under the following cumulative conditions: Links can only be made to webpages that provide a link to the Legal Notice page. | |
| Record name | Ertugliflozin | |
| Source | FDA Global Substance Registration System (GSRS) | |
| URL | https://gsrs.ncats.nih.gov/ginas/app/beta/substances/6C282481IP | |
| Description | The FDA Global Substance Registration System (GSRS) enables the efficient and accurate exchange of information on what substances are in regulated products. Instead of relying on names, which vary across regulatory domains, countries, and regions, the GSRS knowledge base makes it possible for substances to be defined by standardized, scientific descriptions. | |
| Explanation | Unless otherwise noted, the contents of the FDA website (www.fda.gov), both text and graphics, are not copyrighted. They are in the public domain and may be republished, reprinted and otherwise used freely by anyone without the need to obtain permission from FDA. Credit to the U.S. Food and Drug Administration as the source is appreciated but not required. | |
Descargo de responsabilidad e información sobre productos de investigación in vitro
Tenga en cuenta que todos los artículos e información de productos presentados en BenchChem están destinados únicamente con fines informativos. Los productos disponibles para la compra en BenchChem están diseñados específicamente para estudios in vitro, que se realizan fuera de organismos vivos. Los estudios in vitro, derivados del término latino "in vidrio", involucran experimentos realizados en entornos de laboratorio controlados utilizando células o tejidos. Es importante tener en cuenta que estos productos no se clasifican como medicamentos y no han recibido la aprobación de la FDA para la prevención, tratamiento o cura de ninguna condición médica, dolencia o enfermedad. Debemos enfatizar que cualquier forma de introducción corporal de estos productos en humanos o animales está estrictamente prohibida por ley. Es esencial adherirse a estas pautas para garantizar el cumplimiento de los estándares legales y éticos en la investigación y experimentación.



![Sodium ((1,4-phenylenebis(methanylylidene))bis(7,7-dimethyl-2-oxobicyclo[2.2.1]heptan-1-yl-3-ylidene))dimethanesulfonate](/img/structure/B560017.png)
![[[(2R,3R,4R,5R)-5-(6-aminopurin-9-yl)-3-hydroxy-4-phosphonooxyoxolan-2-yl]methoxy-hydroxyphosphoryl] [(2R,3S,4R,5R)-5-(3-carbamoyl-4H-pyridin-1-yl)-3,4-dihydroxyoxolan-2-yl]methyl hydrogen phosphate;cyclohexanamine](/img/structure/B560022.png)
![(R)-3-(4-Phenoxyphenyl)-1-(piperidin-3-yl)-1H-pyrazolo[3,4-d]pyrimidin-4-amine](/img/structure/B560023.png)