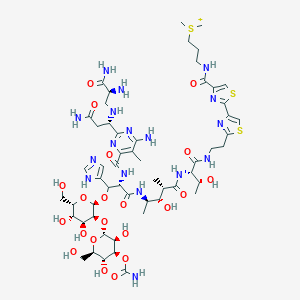
Bleomycine
Vue d'ensemble
Description
Bleomycin is a glycopeptide antibiotic derived from the bacterium Streptomyces verticillus. It is primarily used as an antineoplastic agent in the treatment of various cancers, including Hodgkin’s lymphoma, non-Hodgkin’s lymphoma, testicular cancer, ovarian cancer, and cervical cancer . Bleomycin is known for its ability to induce DNA strand breaks, which makes it effective in cancer therapy .
Mécanisme D'action
Target of Action
Bleomycin, a glycopeptide antibiotic, primarily targets DNA within cells . It selectively inhibits the synthesis of deoxyribonucleic acid (DNA), with some evidence of lesser inhibition of RNA and protein synthesis .
Mode of Action
The primary mechanism by which bleomycin works is by attaching to metal ions like iron and creating metallobleomycin complexes, which causes oxidative damage to DNA . Between 30 and 40 deoxyribose linkages, these reactive oxygen species can disrupt DNA single- and double-strands . This leads to the generation of free base propenals, specifically thymine .
Biochemical Pathways
Bleomycin’s action requires the presence of oxygen and a reducing agent; the metal-containing bleomycin becomes activated and functions as ferrous oxidase . Bleomycin causes oxidative damage to deoxyribose of nucleotides. In particular, the deoxyribose of thymidylate leads to single- and double-stranded breaks in DNA . Multiple DNA breaks lead to cell apoptosis .
Pharmacokinetics
Bleomycin has a half-life of 2 hours . It is 1% protein-bound, with a volume of distribution of 17 L/m² . The metabolism of bleomycin is unknown, but it forms a complex with iron known as the bleomycin-iron complex . It has a clearance rate of 35 mL/min and is excreted in the urine (50-70%) .
Result of Action
Bleomycin exposure induces dose- and time-dependent accumulation of senescence hallmarks and DNA lesions in alveolar epithelial cells (AECs) . These effects are probably due to the inhibition of Rad51 expression, consequently suppressing homologous recombination (HR) repair . Furthermore, the genetic supplement of Rad51 substantially mitigated bleomycin-mediated effects on DSB repair and senescence in AECs .
Action Environment
Risk factors for BPT include decreased glomerular filtration rate, supplementary oxygen, increased creatinine, cumulative dosage, and advanced age . Fever, chest pain, chills, faintness, and shortness of breath are other adverse effects .
Applications De Recherche Scientifique
Bleomycin has a wide range of scientific research applications, particularly in the fields of chemistry, biology, medicine, and industry . In chemistry, bleomycin is used as a model compound to study metal-ion interactions and oxidative DNA damage . In biology, it is used to investigate DNA repair mechanisms and the cellular response to DNA damage . In medicine, bleomycin is a critical component of chemotherapy regimens for various cancers . Additionally, bleomycin has industrial applications in the production of bleomycin analogues and derivatives for research and therapeutic purposes .
Analyse Biochimique
Biochemical Properties
Bleomycin plays a significant role in biochemical reactions. It interacts with various enzymes, proteins, and other biomolecules. The DNA-cleaving actions of Bleomycin are dependent on oxygen and metal ions . It is believed that Bleomycin chelates metal ions (primarily iron) producing a pseudoenzyme that reacts with oxygen to produce superoxide and hydroxide free radicals that cleave DNA .
Cellular Effects
Bleomycin has profound effects on various types of cells and cellular processes. It influences cell function, including impacts on cell signaling pathways, gene expression, and cellular metabolism. The DNA-cleaving actions of Bleomycin can lead to cell death, making it effective as an antitumor antibiotic .
Molecular Mechanism
Bleomycin exerts its effects at the molecular level through several mechanisms. It binds to DNA and induces strand breaks . This binding interaction with DNA, coupled with the production of free radicals, leads to DNA cleavage, which can inhibit DNA synthesis and lead to cell death .
Méthodes De Préparation
Synthetic Routes and Reaction Conditions: Bleomycin is produced through the fermentation of Streptomyces verticillus. The fermentation process involves the cultivation of the bacterium in a nutrient-rich medium, followed by the extraction and purification of the compound . The production of bleomycin involves several steps, including the isolation of the active glycopeptide, its chemical modification, and the formation of the final product .
Industrial Production Methods: Industrial production of bleomycin typically involves large-scale fermentation processes. The bacterium Streptomyces verticillus is grown in bioreactors under controlled conditions to maximize the yield of bleomycin . The fermentation broth is then subjected to various purification steps, including filtration, chromatography, and crystallization, to obtain the pure compound .
Analyse Des Réactions Chimiques
Types of Reactions: Bleomycin undergoes several types of chemical reactions, including oxidation, reduction, and substitution reactions . One notable reaction is the formation of a complex with metal ions, such as iron, which enhances its ability to induce oxidative damage to DNA .
Common Reagents and Conditions: Common reagents used in bleomycin reactions include hydrogen peroxide (H₂O₂) and iron (Fe²⁺) ions . The reaction conditions typically involve the presence of these reagents in an aqueous medium, where bleomycin forms a complex with iron ions, leading to the generation of reactive oxygen species (ROS) that cause DNA strand breaks .
Major Products Formed: The major products formed from bleomycin reactions are DNA fragments resulting from the oxidative cleavage of DNA strands . This cleavage is primarily due to the formation of ROS, which attack the deoxyribose backbone of DNA .
Comparaison Avec Des Composés Similaires
Bleomycin belongs to the glycopeptide antibiotic family, which includes other compounds such as vancomycin and tallysomycin . Compared to these compounds, bleomycin is unique in its ability to induce DNA strand breaks through the formation of metallobleomycin complexes . Vancomycin, for example, primarily targets bacterial cell wall synthesis, while tallysomycin shares a similar mechanism of action with bleomycin but has different clinical applications .
List of Similar Compounds:- Vancomycin
- Tallysomycin
- Calicheamicin
- Neocarzinostatin
Bleomycin’s unique mechanism of action and its effectiveness in cancer therapy make it a valuable compound in both clinical and research settings.
Propriétés
IUPAC Name |
3-[[2-[2-[2-[[(2S,3R)-2-[[(2S,3S,4R)-4-[[(2S,3R)-2-[[6-amino-2-[(1S)-3-amino-1-[[(2S)-2,3-diamino-3-oxopropyl]amino]-3-oxopropyl]-5-methylpyrimidine-4-carbonyl]amino]-3-[3-[4-carbamoyloxy-3,5-dihydroxy-6-(hydroxymethyl)oxan-2-yl]oxy-4,5-dihydroxy-6-(hydroxymethyl)oxan-2-yl]oxy-3-(1H-imidazol-5-yl)propanoyl]amino]-3-hydroxy-2-methylpentanoyl]amino]-3-hydroxybutanoyl]amino]ethyl]-1,3-thiazol-4-yl]-1,3-thiazole-4-carbonyl]amino]propyl-dimethylsulfanium | |
|---|---|---|
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
InChI |
InChI=1S/C55H83N17O21S3/c1-20-33(69-46(72-44(20)58)25(12-31(57)76)64-13-24(56)45(59)82)50(86)71-35(41(26-14-61-19-65-26)91-54-43(39(80)37(78)29(15-73)90-54)92-53-40(81)42(93-55(60)88)38(79)30(16-74)89-53)51(87)66-22(3)36(77)21(2)47(83)70-34(23(4)75)49(85)63-10-8-32-67-28(18-94-32)52-68-27(17-95-52)48(84)62-9-7-11-96(5)6/h14,17-19,21-25,29-30,34-43,53-54,64,73-75,77-81H,7-13,15-16,56H2,1-6H3,(H13-,57,58,59,60,61,62,63,65,66,69,70,71,72,76,82,83,84,85,86,87,88)/p+1/t21-,22+,23+,24-,25-,29?,30?,34-,35-,36-,37?,38?,39?,40?,41-,42?,43?,53?,54?/m0/s1 | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
InChI Key |
OYVAGSVQBOHSSS-WXFSZRTFSA-O | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
Canonical SMILES |
CC1=C(N=C(N=C1N)C(CC(=O)N)NCC(C(=O)N)N)C(=O)NC(C(C2=CN=CN2)OC3C(C(C(C(O3)CO)O)O)OC4C(C(C(C(O4)CO)O)OC(=O)N)O)C(=O)NC(C)C(C(C)C(=O)NC(C(C)O)C(=O)NCCC5=NC(=CS5)C6=NC(=CS6)C(=O)NCCC[S+](C)C)O | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
Isomeric SMILES |
CC1=C(N=C(N=C1N)[C@H](CC(=O)N)NC[C@@H](C(=O)N)N)C(=O)N[C@@H]([C@H](C2=CN=CN2)OC3C(C(C(C(O3)CO)O)O)OC4C(C(C(C(O4)CO)O)OC(=O)N)O)C(=O)N[C@H](C)[C@H]([C@H](C)C(=O)N[C@@H]([C@@H](C)O)C(=O)NCCC5=NC(=CS5)C6=NC(=CS6)C(=O)NCCC[S+](C)C)O | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
Molecular Formula |
C55H84N17O21S3+ | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
Related CAS |
9041-93-4 (sulfate (salt)) | |
| Record name | Bleomycin [INN:BAN] | |
| Source | ChemIDplus | |
| URL | https://pubchem.ncbi.nlm.nih.gov/substance/?source=chemidplus&sourceid=0011056067 | |
| Description | ChemIDplus is a free, web search system that provides access to the structure and nomenclature authority files used for the identification of chemical substances cited in National Library of Medicine (NLM) databases, including the TOXNET system. | |
Molecular Weight |
1415.6 g/mol | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
Physical Description |
Bleomycin appears as colorless or yellowish powder. Possible bluish color depending on copper content. (NTP, 1992), Solid | |
| Record name | BLEOMYCIN | |
| Source | CAMEO Chemicals | |
| URL | https://cameochemicals.noaa.gov/chemical/19884 | |
| Description | CAMEO Chemicals is a chemical database designed for people who are involved in hazardous material incident response and planning. CAMEO Chemicals contains a library with thousands of datasheets containing response-related information and recommendations for hazardous materials that are commonly transported, used, or stored in the United States. CAMEO Chemicals was developed by the National Oceanic and Atmospheric Administration's Office of Response and Restoration in partnership with the Environmental Protection Agency's Office of Emergency Management. | |
| Explanation | CAMEO Chemicals and all other CAMEO products are available at no charge to those organizations and individuals (recipients) responsible for the safe handling of chemicals. However, some of the chemical data itself is subject to the copyright restrictions of the companies or organizations that provided the data. | |
| Record name | Bleomycin | |
| Source | Human Metabolome Database (HMDB) | |
| URL | http://www.hmdb.ca/metabolites/HMDB0014435 | |
| Description | The Human Metabolome Database (HMDB) is a freely available electronic database containing detailed information about small molecule metabolites found in the human body. | |
| Explanation | HMDB is offered to the public as a freely available resource. Use and re-distribution of the data, in whole or in part, for commercial purposes requires explicit permission of the authors and explicit acknowledgment of the source material (HMDB) and the original publication (see the HMDB citing page). We ask that users who download significant portions of the database cite the HMDB paper in any resulting publications. | |
Solubility |
Soluble (NTP, 1992), Soluble, Colorless or yellowish powder which becomes bluish depending on copper content. Very sol in water, methanol; practically insol in acetone, ethyl acetate, butyl acetate, ether; slightly sol in ethanol. /Bleomycins/, HIGHLY SOL IN WATER & METHANOL; SPARINGLY SOL IN ALC; INSOL IN ACETONE & ETHYL ACETATE; CREAM-COLORED POWDER OR SOFT, FLUFFY LUMPS. /SULFATE SALT/, Freely soluble in water., Sol in water and methanol but insol in acetone and ether., 2.82e-02 g/L | |
| Record name | BLEOMYCIN | |
| Source | CAMEO Chemicals | |
| URL | https://cameochemicals.noaa.gov/chemical/19884 | |
| Description | CAMEO Chemicals is a chemical database designed for people who are involved in hazardous material incident response and planning. CAMEO Chemicals contains a library with thousands of datasheets containing response-related information and recommendations for hazardous materials that are commonly transported, used, or stored in the United States. CAMEO Chemicals was developed by the National Oceanic and Atmospheric Administration's Office of Response and Restoration in partnership with the Environmental Protection Agency's Office of Emergency Management. | |
| Explanation | CAMEO Chemicals and all other CAMEO products are available at no charge to those organizations and individuals (recipients) responsible for the safe handling of chemicals. However, some of the chemical data itself is subject to the copyright restrictions of the companies or organizations that provided the data. | |
| Record name | Bleomycin | |
| Source | DrugBank | |
| URL | https://www.drugbank.ca/drugs/DB00290 | |
| Description | The DrugBank database is a unique bioinformatics and cheminformatics resource that combines detailed drug (i.e. chemical, pharmacological and pharmaceutical) data with comprehensive drug target (i.e. sequence, structure, and pathway) information. | |
| Explanation | Creative Common's Attribution-NonCommercial 4.0 International License (http://creativecommons.org/licenses/by-nc/4.0/legalcode) | |
| Record name | BLEOMYCIN | |
| Source | Hazardous Substances Data Bank (HSDB) | |
| URL | https://pubchem.ncbi.nlm.nih.gov/source/hsdb/3208 | |
| Description | The Hazardous Substances Data Bank (HSDB) is a toxicology database that focuses on the toxicology of potentially hazardous chemicals. It provides information on human exposure, industrial hygiene, emergency handling procedures, environmental fate, regulatory requirements, nanomaterials, and related areas. The information in HSDB has been assessed by a Scientific Review Panel. | |
| Record name | Bleomycin | |
| Source | Human Metabolome Database (HMDB) | |
| URL | http://www.hmdb.ca/metabolites/HMDB0014435 | |
| Description | The Human Metabolome Database (HMDB) is a freely available electronic database containing detailed information about small molecule metabolites found in the human body. | |
| Explanation | HMDB is offered to the public as a freely available resource. Use and re-distribution of the data, in whole or in part, for commercial purposes requires explicit permission of the authors and explicit acknowledgment of the source material (HMDB) and the original publication (see the HMDB citing page). We ask that users who download significant portions of the database cite the HMDB paper in any resulting publications. | |
Mechanism of Action |
Although the exact mechanism of action of bleomycin is unknown, available evidence would seem to indicate that the main mode of action is the inhibition of DNA synthesis with some evidence of lesser inhibition of RNA and protein synthesis. As evident in _in vitro_ studies, the DNA-cleaving actions of bleomycin is dependent on oxygen and metal ions. It is believed that bleomycin chelates metal ions (primarily iron) producing a pseudoenzyme that reacts with oxygen to produce superoxide and hydroxide free radicals that cleave DNA., Cytotoxic action of bleomycins results from their ability to cause fragmentation of DNA. Studies in vitro indicate that bleomycin causes accumulation of cells in the G2 phase of the cell cycle, and many of these cells display chromosomal aberrations, incl chromatid breaks, gaps, and fragments, as well as translocations. Bleomycin appears to cause scission of DNA by interacting with oxygen and iron(2+). In the presence of oxygen and a reducing agent, such as dithiothreitol, the metallobleomycin complex becomes activated and functions mechanistically as a ferrous oxidase, transferring electrons from iron(2) to molecular oxygen to produce activated species of oxygen. It has also been shown that metallobleomycin complexes can be activated by reaction with the flavin enzyme, NADPH-cytochrome p450 reductase. Bleomycin binds to DNA through its amino terminal peptide, and the activated complex generates free radicals that are responsible for scission of the DNA chain., Bleomycin is an antineoplastic antibiotic. The drug is active against gram-positive and gram-negative bacteria and fungi, but its cytotoxicity precludes its use as an anti-infective agent. The precise mechanism(s) of action of bleomycin is not fully known. Several studies in Escherichia coli and HeLa cells suggest that the drug inhibits the incorporation of thymidine into DNA. In these in vitro studies, DNA synthesis was inhibited to a greater extent than was RNA or protein synthesis. Bleomycin also appears to labilize the DNA structure, resulting in scission of both single- and double-stranded DNA. The drug has no immunosuppressive activity in mice., Bleomycin is classed as an antibiotic but is not used as an antimicrobial agent. Although bleomycin is effective against both cycling and non-cycling cells, it seems to be most effective in the G2 phase of cell division. Its exact mechanism of antineoplastic action is unknown but may involve binding to DNA, inducing lability of the DNA structure, and reduced synthesis of DNA, and to a lesser extent RNA and ptoteins., When administered into the pleural cavity in the treatment of malignant pleural effusion, /bleomycin/ acts as a sclerosing agent. | |
| Record name | Bleomycin | |
| Source | DrugBank | |
| URL | https://www.drugbank.ca/drugs/DB00290 | |
| Description | The DrugBank database is a unique bioinformatics and cheminformatics resource that combines detailed drug (i.e. chemical, pharmacological and pharmaceutical) data with comprehensive drug target (i.e. sequence, structure, and pathway) information. | |
| Explanation | Creative Common's Attribution-NonCommercial 4.0 International License (http://creativecommons.org/licenses/by-nc/4.0/legalcode) | |
| Record name | BLEOMYCIN | |
| Source | Hazardous Substances Data Bank (HSDB) | |
| URL | https://pubchem.ncbi.nlm.nih.gov/source/hsdb/3208 | |
| Description | The Hazardous Substances Data Bank (HSDB) is a toxicology database that focuses on the toxicology of potentially hazardous chemicals. It provides information on human exposure, industrial hygiene, emergency handling procedures, environmental fate, regulatory requirements, nanomaterials, and related areas. The information in HSDB has been assessed by a Scientific Review Panel. | |
Color/Form |
Colorless to yellow powder | |
CAS No. |
11056-06-7 | |
| Record name | BLEOMYCIN | |
| Source | CAMEO Chemicals | |
| URL | https://cameochemicals.noaa.gov/chemical/19884 | |
| Description | CAMEO Chemicals is a chemical database designed for people who are involved in hazardous material incident response and planning. CAMEO Chemicals contains a library with thousands of datasheets containing response-related information and recommendations for hazardous materials that are commonly transported, used, or stored in the United States. CAMEO Chemicals was developed by the National Oceanic and Atmospheric Administration's Office of Response and Restoration in partnership with the Environmental Protection Agency's Office of Emergency Management. | |
| Explanation | CAMEO Chemicals and all other CAMEO products are available at no charge to those organizations and individuals (recipients) responsible for the safe handling of chemicals. However, some of the chemical data itself is subject to the copyright restrictions of the companies or organizations that provided the data. | |
| Record name | Bleomycin [INN:BAN] | |
| Source | ChemIDplus | |
| URL | https://pubchem.ncbi.nlm.nih.gov/substance/?source=chemidplus&sourceid=0011056067 | |
| Description | ChemIDplus is a free, web search system that provides access to the structure and nomenclature authority files used for the identification of chemical substances cited in National Library of Medicine (NLM) databases, including the TOXNET system. | |
| Record name | Bleomycin | |
| Source | DrugBank | |
| URL | https://www.drugbank.ca/drugs/DB00290 | |
| Description | The DrugBank database is a unique bioinformatics and cheminformatics resource that combines detailed drug (i.e. chemical, pharmacological and pharmaceutical) data with comprehensive drug target (i.e. sequence, structure, and pathway) information. | |
| Explanation | Creative Common's Attribution-NonCommercial 4.0 International License (http://creativecommons.org/licenses/by-nc/4.0/legalcode) | |
| Record name | BLEOMYCIN | |
| Source | Hazardous Substances Data Bank (HSDB) | |
| URL | https://pubchem.ncbi.nlm.nih.gov/source/hsdb/3208 | |
| Description | The Hazardous Substances Data Bank (HSDB) is a toxicology database that focuses on the toxicology of potentially hazardous chemicals. It provides information on human exposure, industrial hygiene, emergency handling procedures, environmental fate, regulatory requirements, nanomaterials, and related areas. The information in HSDB has been assessed by a Scientific Review Panel. | |
| Record name | Bleomycin | |
| Source | Human Metabolome Database (HMDB) | |
| URL | http://www.hmdb.ca/metabolites/HMDB0014435 | |
| Description | The Human Metabolome Database (HMDB) is a freely available electronic database containing detailed information about small molecule metabolites found in the human body. | |
| Explanation | HMDB is offered to the public as a freely available resource. Use and re-distribution of the data, in whole or in part, for commercial purposes requires explicit permission of the authors and explicit acknowledgment of the source material (HMDB) and the original publication (see the HMDB citing page). We ask that users who download significant portions of the database cite the HMDB paper in any resulting publications. | |
Melting Point |
71 °C | |
| Record name | Bleomycin | |
| Source | DrugBank | |
| URL | https://www.drugbank.ca/drugs/DB00290 | |
| Description | The DrugBank database is a unique bioinformatics and cheminformatics resource that combines detailed drug (i.e. chemical, pharmacological and pharmaceutical) data with comprehensive drug target (i.e. sequence, structure, and pathway) information. | |
| Explanation | Creative Common's Attribution-NonCommercial 4.0 International License (http://creativecommons.org/licenses/by-nc/4.0/legalcode) | |
| Record name | Bleomycin | |
| Source | Human Metabolome Database (HMDB) | |
| URL | http://www.hmdb.ca/metabolites/HMDB0014435 | |
| Description | The Human Metabolome Database (HMDB) is a freely available electronic database containing detailed information about small molecule metabolites found in the human body. | |
| Explanation | HMDB is offered to the public as a freely available resource. Use and re-distribution of the data, in whole or in part, for commercial purposes requires explicit permission of the authors and explicit acknowledgment of the source material (HMDB) and the original publication (see the HMDB citing page). We ask that users who download significant portions of the database cite the HMDB paper in any resulting publications. | |
Retrosynthesis Analysis
AI-Powered Synthesis Planning: Our tool employs the Template_relevance Pistachio, Template_relevance Bkms_metabolic, Template_relevance Pistachio_ringbreaker, Template_relevance Reaxys, Template_relevance Reaxys_biocatalysis model, leveraging a vast database of chemical reactions to predict feasible synthetic routes.
One-Step Synthesis Focus: Specifically designed for one-step synthesis, it provides concise and direct routes for your target compounds, streamlining the synthesis process.
Accurate Predictions: Utilizing the extensive PISTACHIO, BKMS_METABOLIC, PISTACHIO_RINGBREAKER, REAXYS, REAXYS_BIOCATALYSIS database, our tool offers high-accuracy predictions, reflecting the latest in chemical research and data.
Strategy Settings
| Precursor scoring | Relevance Heuristic |
|---|---|
| Min. plausibility | 0.01 |
| Model | Template_relevance |
| Template Set | Pistachio/Bkms_metabolic/Pistachio_ringbreaker/Reaxys/Reaxys_biocatalysis |
| Top-N result to add to graph | 6 |
Feasible Synthetic Routes
Avertissement et informations sur les produits de recherche in vitro
Veuillez noter que tous les articles et informations sur les produits présentés sur BenchChem sont destinés uniquement à des fins informatives. Les produits disponibles à l'achat sur BenchChem sont spécifiquement conçus pour des études in vitro, qui sont réalisées en dehors des organismes vivants. Les études in vitro, dérivées du terme latin "in verre", impliquent des expériences réalisées dans des environnements de laboratoire contrôlés à l'aide de cellules ou de tissus. Il est important de noter que ces produits ne sont pas classés comme médicaments et n'ont pas reçu l'approbation de la FDA pour la prévention, le traitement ou la guérison de toute condition médicale, affection ou maladie. Nous devons souligner que toute forme d'introduction corporelle de ces produits chez les humains ou les animaux est strictement interdite par la loi. Il est essentiel de respecter ces directives pour assurer la conformité aux normes légales et éthiques en matière de recherche et d'expérimentation.


![8-Methylthieno[2,3-g][1,3]benzothiazol-2-amine](/img/structure/B88125.png)