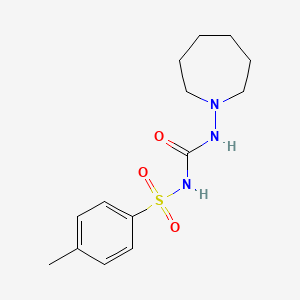
Tolazamide
Vue d'ensemble
Description
Tolazamide is an oral blood glucose-lowering drug used primarily for the treatment of Type 2 diabetes mellitus. It belongs to the sulfonylurea class of medications, which function by stimulating the release of insulin from the pancreas. This compound is particularly effective in patients whose hyperglycemia cannot be controlled by diet alone .
Mécanisme D'action
Target of Action
Tolazamide is a sulfonylurea class drug that primarily targets the beta cells in the pancreatic islets . These cells play a crucial role in the regulation of blood glucose levels by producing and releasing insulin, a hormone that facilitates the uptake of glucose by cells.
Mode of Action
This compound acts by stimulating the release of insulin from the pancreas . This effect is dependent on the functioning of the beta cells in the pancreatic islets . By increasing the release of insulin, this compound enhances the body’s ability to lower blood glucose levels.
Pharmacokinetics
This compound is well absorbed from the gastrointestinal tract . It is extensively metabolized in the liver to five major metabolites, with hypoglycemic activity ranging from 0% to 70% . The drug is excreted primarily in the urine (approximately 85%) and to a lesser extent in the feces (about 7%) . The elimination half-life of this compound is approximately 7 hours .
Result of Action
The primary result of this compound’s action is a reduction in blood glucose levels. By stimulating the release of insulin from the pancreas, this compound facilitates the uptake of glucose by cells, thereby lowering blood glucose levels .
Applications De Recherche Scientifique
Tolazamide has a wide range of scientific research applications:
Chemistry: It is used as a model compound in studies of sulfonylurea chemistry and reaction mechanisms.
Biology: this compound is studied for its effects on cellular metabolism and insulin secretion.
Medicine: It is extensively researched for its therapeutic potential in managing Type 2 diabetes mellitus.
Industry: this compound is used in the pharmaceutical industry for the development of new antidiabetic drugs.
Analyse Biochimique
Biochemical Properties
Tolazamide interacts with various enzymes and proteins to exert its effects. It appears to lower the blood glucose acutely by stimulating the release of insulin from the pancreas, an effect dependent upon functioning beta cells in the pancreatic islets .
Cellular Effects
This compound influences cell function by lowering blood glucose levels. This is achieved by stimulating the release of insulin from the pancreas . The mechanism by which this compound lowers blood glucose during long-term administration has not been clearly established .
Molecular Mechanism
This compound exerts its effects at the molecular level by stimulating the release of insulin from the pancreas . This effect is dependent upon functioning beta cells in the pancreatic islets . The exact mechanism by which this compound lowers blood glucose during long-term administration has not been clearly established .
Temporal Effects in Laboratory Settings
With chronic administration in Type II diabetic patients, the blood glucose-lowering effect of this compound persists despite a gradual decline in the insulin secretory response to the drug . The average biological half-life of the drug is 7 hours .
Metabolic Pathways
This compound is metabolized in the liver to five major metabolites ranging in hypoglycemic activity from 0% to 70% . They are excreted principally in the urine .
Méthodes De Préparation
Synthetic Routes and Reaction Conditions: Tolazamide is synthesized through a multi-step process. The synthesis begins with the conversion of para-toluenesulfonamide to its carbamate using ethyl chloroformate in the presence of a base. This intermediate is then heated with 1-amino-azepane, leading to the displacement of the ethoxy group and the formation of this compound .
Industrial Production Methods: In industrial settings, the production of this compound involves similar synthetic routes but on a larger scale. The process is optimized for yield and purity, ensuring that the final product meets pharmaceutical standards. The reaction conditions are carefully controlled to maintain consistency and efficiency .
Analyse Des Réactions Chimiques
Types of Reactions: Tolazamide undergoes various chemical reactions, including:
Oxidation: this compound can be oxidized under specific conditions, leading to the formation of sulfoxides and sulfones.
Reduction: Reduction reactions can convert this compound to its corresponding amine derivatives.
Substitution: this compound can undergo nucleophilic substitution reactions, particularly at the sulfonylurea moiety.
Common Reagents and Conditions:
Oxidation: Common oxidizing agents include hydrogen peroxide and potassium permanganate.
Reduction: Reducing agents such as lithium aluminum hydride and sodium borohydride are used.
Substitution: Nucleophiles like amines and thiols are commonly employed in substitution reactions.
Major Products: The major products formed from these reactions include sulfoxides, sulfones, amine derivatives, and substituted sulfonylureas .
Comparaison Avec Des Composés Similaires
- Chlorpropamide
- Glipizide
- Glyburide
- Glimepiride
Comparison: Tolazamide is unique among sulfonylureas due to its specific pharmacokinetic properties, including its relatively long half-life and its ability to produce a mild diuretic effect by enhancing renal free water clearance. Compared to other sulfonylureas, this compound has a more gradual onset of action and a longer duration of effect, making it suitable for patients who require sustained blood glucose control .
Propriétés
IUPAC Name |
1-(azepan-1-yl)-3-(4-methylphenyl)sulfonylurea | |
|---|---|---|
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
InChI |
InChI=1S/C14H21N3O3S/c1-12-6-8-13(9-7-12)21(19,20)16-14(18)15-17-10-4-2-3-5-11-17/h6-9H,2-5,10-11H2,1H3,(H2,15,16,18) | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
InChI Key |
OUDSBRTVNLOZBN-UHFFFAOYSA-N | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
Canonical SMILES |
CC1=CC=C(C=C1)S(=O)(=O)NC(=O)NN2CCCCCC2 | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
Molecular Formula |
C14H21N3O3S | |
| Record name | TOLAZAMIDE | |
| Source | CAMEO Chemicals | |
| URL | https://cameochemicals.noaa.gov/chemical/21119 | |
| Description | CAMEO Chemicals is a chemical database designed for people who are involved in hazardous material incident response and planning. CAMEO Chemicals contains a library with thousands of datasheets containing response-related information and recommendations for hazardous materials that are commonly transported, used, or stored in the United States. CAMEO Chemicals was developed by the National Oceanic and Atmospheric Administration's Office of Response and Restoration in partnership with the Environmental Protection Agency's Office of Emergency Management. | |
| Explanation | CAMEO Chemicals and all other CAMEO products are available at no charge to those organizations and individuals (recipients) responsible for the safe handling of chemicals. However, some of the chemical data itself is subject to the copyright restrictions of the companies or organizations that provided the data. | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
DSSTOX Substance ID |
DTXSID3021358 | |
| Record name | Tolazamide | |
| Source | EPA DSSTox | |
| URL | https://comptox.epa.gov/dashboard/DTXSID3021358 | |
| Description | DSSTox provides a high quality public chemistry resource for supporting improved predictive toxicology. | |
Molecular Weight |
311.40 g/mol | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
Physical Description |
Tolazamide appears as white to off-white crystalline powder. Odorless or with a slight odor. (NTP, 1992), Solid | |
| Record name | TOLAZAMIDE | |
| Source | CAMEO Chemicals | |
| URL | https://cameochemicals.noaa.gov/chemical/21119 | |
| Description | CAMEO Chemicals is a chemical database designed for people who are involved in hazardous material incident response and planning. CAMEO Chemicals contains a library with thousands of datasheets containing response-related information and recommendations for hazardous materials that are commonly transported, used, or stored in the United States. CAMEO Chemicals was developed by the National Oceanic and Atmospheric Administration's Office of Response and Restoration in partnership with the Environmental Protection Agency's Office of Emergency Management. | |
| Explanation | CAMEO Chemicals and all other CAMEO products are available at no charge to those organizations and individuals (recipients) responsible for the safe handling of chemicals. However, some of the chemical data itself is subject to the copyright restrictions of the companies or organizations that provided the data. | |
| Record name | Tolazamide | |
| Source | Human Metabolome Database (HMDB) | |
| URL | http://www.hmdb.ca/metabolites/HMDB0014977 | |
| Description | The Human Metabolome Database (HMDB) is a freely available electronic database containing detailed information about small molecule metabolites found in the human body. | |
| Explanation | HMDB is offered to the public as a freely available resource. Use and re-distribution of the data, in whole or in part, for commercial purposes requires explicit permission of the authors and explicit acknowledgment of the source material (HMDB) and the original publication (see the HMDB citing page). We ask that users who download significant portions of the database cite the HMDB paper in any resulting publications. | |
Solubility |
less than 1 mg/mL at 77 °F (NTP, 1992), VERY SLIGHTLY SOL IN WATER; FREELY SOL IN CHLOROFORM; SOL IN ACETONE; SIGHTLY SOL IN ALCOHOL, In water, 65.4 mg/l @ 30 °C, 3.08e-01 g/L | |
| Record name | TOLAZAMIDE | |
| Source | CAMEO Chemicals | |
| URL | https://cameochemicals.noaa.gov/chemical/21119 | |
| Description | CAMEO Chemicals is a chemical database designed for people who are involved in hazardous material incident response and planning. CAMEO Chemicals contains a library with thousands of datasheets containing response-related information and recommendations for hazardous materials that are commonly transported, used, or stored in the United States. CAMEO Chemicals was developed by the National Oceanic and Atmospheric Administration's Office of Response and Restoration in partnership with the Environmental Protection Agency's Office of Emergency Management. | |
| Explanation | CAMEO Chemicals and all other CAMEO products are available at no charge to those organizations and individuals (recipients) responsible for the safe handling of chemicals. However, some of the chemical data itself is subject to the copyright restrictions of the companies or organizations that provided the data. | |
| Record name | Tolazamide | |
| Source | DrugBank | |
| URL | https://www.drugbank.ca/drugs/DB00839 | |
| Description | The DrugBank database is a unique bioinformatics and cheminformatics resource that combines detailed drug (i.e. chemical, pharmacological and pharmaceutical) data with comprehensive drug target (i.e. sequence, structure, and pathway) information. | |
| Explanation | Creative Common's Attribution-NonCommercial 4.0 International License (http://creativecommons.org/licenses/by-nc/4.0/legalcode) | |
| Record name | TOLAZAMIDE | |
| Source | Hazardous Substances Data Bank (HSDB) | |
| URL | https://pubchem.ncbi.nlm.nih.gov/source/hsdb/3192 | |
| Description | The Hazardous Substances Data Bank (HSDB) is a toxicology database that focuses on the toxicology of potentially hazardous chemicals. It provides information on human exposure, industrial hygiene, emergency handling procedures, environmental fate, regulatory requirements, nanomaterials, and related areas. The information in HSDB has been assessed by a Scientific Review Panel. | |
| Record name | Tolazamide | |
| Source | Human Metabolome Database (HMDB) | |
| URL | http://www.hmdb.ca/metabolites/HMDB0014977 | |
| Description | The Human Metabolome Database (HMDB) is a freely available electronic database containing detailed information about small molecule metabolites found in the human body. | |
| Explanation | HMDB is offered to the public as a freely available resource. Use and re-distribution of the data, in whole or in part, for commercial purposes requires explicit permission of the authors and explicit acknowledgment of the source material (HMDB) and the original publication (see the HMDB citing page). We ask that users who download significant portions of the database cite the HMDB paper in any resulting publications. | |
Mechanism of Action |
Sulfonylureas likely bind to ATP-sensitive potassium-channel receptors on the pancreatic cell surface, reducing potassium conductance and causing depolarization of the membrane. Depolarization stimulates calcium ion influx through voltage-sensitive calcium channels, raising intracellular concentrations of calcium ions, which induces the secretion, or exocytosis, of insulin., Sulfonylureas are now...thought to act by a number of different mechanisms. 1. ...produce a depolarization of the pancreatic islet beta cell membrane potassium ion permeability. This results in a release of preformed insulin into the circulation and occurs mostly in non-insulin dependent diabetics. 2. ...reduce basal glucose output from the liver... 3. increase insulin receptor binding... 4. ...increasing intracellular levels of AMP... 5. increase insulin secretion by suppressing the release of glucagon and somatostatin from alpha and delta pancreatic cells. /Sulfonylureas/, Sulfonylureas lower blood glucose in NIDDM by directly stimulating the acute release of insulin from functioning beta cells of pancreatic islet tissue by an unknown process that involves a sulfonylurea receptor on the beta cell. Sulfonylureas inhibit the ATP potassium channels on the beta cell membrane and potassium efflux, which results in depolarization and calcium influx, calcium-calmodulin binding, kinase activation, and release of insulin containing granules by exocytosis, an effect similar to that of glucose. Insulin is a hormone that lowers blood glucose and controls the storage and metabolism of carbohydrates, proteins, and fats. Therefore, sulfonylureas are effective only in patients whose pancreata are capable of producing insulin. /Sulfonylurea antidiabetic agents/ | |
| Record name | Tolazamide | |
| Source | DrugBank | |
| URL | https://www.drugbank.ca/drugs/DB00839 | |
| Description | The DrugBank database is a unique bioinformatics and cheminformatics resource that combines detailed drug (i.e. chemical, pharmacological and pharmaceutical) data with comprehensive drug target (i.e. sequence, structure, and pathway) information. | |
| Explanation | Creative Common's Attribution-NonCommercial 4.0 International License (http://creativecommons.org/licenses/by-nc/4.0/legalcode) | |
| Record name | TOLAZAMIDE | |
| Source | Hazardous Substances Data Bank (HSDB) | |
| URL | https://pubchem.ncbi.nlm.nih.gov/source/hsdb/3192 | |
| Description | The Hazardous Substances Data Bank (HSDB) is a toxicology database that focuses on the toxicology of potentially hazardous chemicals. It provides information on human exposure, industrial hygiene, emergency handling procedures, environmental fate, regulatory requirements, nanomaterials, and related areas. The information in HSDB has been assessed by a Scientific Review Panel. | |
Color/Form |
Crystals, WHITE TO OFF-WHITE, CRYSTALLINE POWDER | |
CAS No. |
1156-19-0 | |
| Record name | TOLAZAMIDE | |
| Source | CAMEO Chemicals | |
| URL | https://cameochemicals.noaa.gov/chemical/21119 | |
| Description | CAMEO Chemicals is a chemical database designed for people who are involved in hazardous material incident response and planning. CAMEO Chemicals contains a library with thousands of datasheets containing response-related information and recommendations for hazardous materials that are commonly transported, used, or stored in the United States. CAMEO Chemicals was developed by the National Oceanic and Atmospheric Administration's Office of Response and Restoration in partnership with the Environmental Protection Agency's Office of Emergency Management. | |
| Explanation | CAMEO Chemicals and all other CAMEO products are available at no charge to those organizations and individuals (recipients) responsible for the safe handling of chemicals. However, some of the chemical data itself is subject to the copyright restrictions of the companies or organizations that provided the data. | |
| Record name | Tolazamide | |
| Source | CAS Common Chemistry | |
| URL | https://commonchemistry.cas.org/detail?cas_rn=1156-19-0 | |
| Description | CAS Common Chemistry is an open community resource for accessing chemical information. Nearly 500,000 chemical substances from CAS REGISTRY cover areas of community interest, including common and frequently regulated chemicals, and those relevant to high school and undergraduate chemistry classes. This chemical information, curated by our expert scientists, is provided in alignment with our mission as a division of the American Chemical Society. | |
| Explanation | The data from CAS Common Chemistry is provided under a CC-BY-NC 4.0 license, unless otherwise stated. | |
| Record name | Tolazamide [USAN:USP:INN:BAN:JAN] | |
| Source | ChemIDplus | |
| URL | https://pubchem.ncbi.nlm.nih.gov/substance/?source=chemidplus&sourceid=0001156190 | |
| Description | ChemIDplus is a free, web search system that provides access to the structure and nomenclature authority files used for the identification of chemical substances cited in National Library of Medicine (NLM) databases, including the TOXNET system. | |
| Record name | Tolazamide | |
| Source | DrugBank | |
| URL | https://www.drugbank.ca/drugs/DB00839 | |
| Description | The DrugBank database is a unique bioinformatics and cheminformatics resource that combines detailed drug (i.e. chemical, pharmacological and pharmaceutical) data with comprehensive drug target (i.e. sequence, structure, and pathway) information. | |
| Explanation | Creative Common's Attribution-NonCommercial 4.0 International License (http://creativecommons.org/licenses/by-nc/4.0/legalcode) | |
| Record name | tolazamide | |
| Source | DTP/NCI | |
| URL | https://dtp.cancer.gov/dtpstandard/servlet/dwindex?searchtype=NSC&outputformat=html&searchlist=758149 | |
| Description | The NCI Development Therapeutics Program (DTP) provides services and resources to the academic and private-sector research communities worldwide to facilitate the discovery and development of new cancer therapeutic agents. | |
| Explanation | Unless otherwise indicated, all text within NCI products is free of copyright and may be reused without our permission. Credit the National Cancer Institute as the source. | |
| Record name | tolazamide | |
| Source | DTP/NCI | |
| URL | https://dtp.cancer.gov/dtpstandard/servlet/dwindex?searchtype=NSC&outputformat=html&searchlist=70762 | |
| Description | The NCI Development Therapeutics Program (DTP) provides services and resources to the academic and private-sector research communities worldwide to facilitate the discovery and development of new cancer therapeutic agents. | |
| Explanation | Unless otherwise indicated, all text within NCI products is free of copyright and may be reused without our permission. Credit the National Cancer Institute as the source. | |
| Record name | Tolazamide | |
| Source | EPA DSSTox | |
| URL | https://comptox.epa.gov/dashboard/DTXSID3021358 | |
| Description | DSSTox provides a high quality public chemistry resource for supporting improved predictive toxicology. | |
| Record name | Tolazamide | |
| Source | European Chemicals Agency (ECHA) | |
| URL | https://echa.europa.eu/substance-information/-/substanceinfo/100.013.262 | |
| Description | The European Chemicals Agency (ECHA) is an agency of the European Union which is the driving force among regulatory authorities in implementing the EU's groundbreaking chemicals legislation for the benefit of human health and the environment as well as for innovation and competitiveness. | |
| Explanation | Use of the information, documents and data from the ECHA website is subject to the terms and conditions of this Legal Notice, and subject to other binding limitations provided for under applicable law, the information, documents and data made available on the ECHA website may be reproduced, distributed and/or used, totally or in part, for non-commercial purposes provided that ECHA is acknowledged as the source: "Source: European Chemicals Agency, http://echa.europa.eu/". Such acknowledgement must be included in each copy of the material. ECHA permits and encourages organisations and individuals to create links to the ECHA website under the following cumulative conditions: Links can only be made to webpages that provide a link to the Legal Notice page. | |
| Record name | TOLAZAMIDE | |
| Source | FDA Global Substance Registration System (GSRS) | |
| URL | https://gsrs.ncats.nih.gov/ginas/app/beta/substances/9LT1BRO48Q | |
| Description | The FDA Global Substance Registration System (GSRS) enables the efficient and accurate exchange of information on what substances are in regulated products. Instead of relying on names, which vary across regulatory domains, countries, and regions, the GSRS knowledge base makes it possible for substances to be defined by standardized, scientific descriptions. | |
| Explanation | Unless otherwise noted, the contents of the FDA website (www.fda.gov), both text and graphics, are not copyrighted. They are in the public domain and may be republished, reprinted and otherwise used freely by anyone without the need to obtain permission from FDA. Credit to the U.S. Food and Drug Administration as the source is appreciated but not required. | |
| Record name | TOLAZAMIDE | |
| Source | Hazardous Substances Data Bank (HSDB) | |
| URL | https://pubchem.ncbi.nlm.nih.gov/source/hsdb/3192 | |
| Description | The Hazardous Substances Data Bank (HSDB) is a toxicology database that focuses on the toxicology of potentially hazardous chemicals. It provides information on human exposure, industrial hygiene, emergency handling procedures, environmental fate, regulatory requirements, nanomaterials, and related areas. The information in HSDB has been assessed by a Scientific Review Panel. | |
| Record name | Tolazamide | |
| Source | Human Metabolome Database (HMDB) | |
| URL | http://www.hmdb.ca/metabolites/HMDB0014977 | |
| Description | The Human Metabolome Database (HMDB) is a freely available electronic database containing detailed information about small molecule metabolites found in the human body. | |
| Explanation | HMDB is offered to the public as a freely available resource. Use and re-distribution of the data, in whole or in part, for commercial purposes requires explicit permission of the authors and explicit acknowledgment of the source material (HMDB) and the original publication (see the HMDB citing page). We ask that users who download significant portions of the database cite the HMDB paper in any resulting publications. | |
Melting Point |
322 to 336 °F (with decomposition) (NTP, 1992), 170-173 °C, 170 - 173 °C | |
| Record name | TOLAZAMIDE | |
| Source | CAMEO Chemicals | |
| URL | https://cameochemicals.noaa.gov/chemical/21119 | |
| Description | CAMEO Chemicals is a chemical database designed for people who are involved in hazardous material incident response and planning. CAMEO Chemicals contains a library with thousands of datasheets containing response-related information and recommendations for hazardous materials that are commonly transported, used, or stored in the United States. CAMEO Chemicals was developed by the National Oceanic and Atmospheric Administration's Office of Response and Restoration in partnership with the Environmental Protection Agency's Office of Emergency Management. | |
| Explanation | CAMEO Chemicals and all other CAMEO products are available at no charge to those organizations and individuals (recipients) responsible for the safe handling of chemicals. However, some of the chemical data itself is subject to the copyright restrictions of the companies or organizations that provided the data. | |
| Record name | Tolazamide | |
| Source | DrugBank | |
| URL | https://www.drugbank.ca/drugs/DB00839 | |
| Description | The DrugBank database is a unique bioinformatics and cheminformatics resource that combines detailed drug (i.e. chemical, pharmacological and pharmaceutical) data with comprehensive drug target (i.e. sequence, structure, and pathway) information. | |
| Explanation | Creative Common's Attribution-NonCommercial 4.0 International License (http://creativecommons.org/licenses/by-nc/4.0/legalcode) | |
| Record name | TOLAZAMIDE | |
| Source | Hazardous Substances Data Bank (HSDB) | |
| URL | https://pubchem.ncbi.nlm.nih.gov/source/hsdb/3192 | |
| Description | The Hazardous Substances Data Bank (HSDB) is a toxicology database that focuses on the toxicology of potentially hazardous chemicals. It provides information on human exposure, industrial hygiene, emergency handling procedures, environmental fate, regulatory requirements, nanomaterials, and related areas. The information in HSDB has been assessed by a Scientific Review Panel. | |
| Record name | Tolazamide | |
| Source | Human Metabolome Database (HMDB) | |
| URL | http://www.hmdb.ca/metabolites/HMDB0014977 | |
| Description | The Human Metabolome Database (HMDB) is a freely available electronic database containing detailed information about small molecule metabolites found in the human body. | |
| Explanation | HMDB is offered to the public as a freely available resource. Use and re-distribution of the data, in whole or in part, for commercial purposes requires explicit permission of the authors and explicit acknowledgment of the source material (HMDB) and the original publication (see the HMDB citing page). We ask that users who download significant portions of the database cite the HMDB paper in any resulting publications. | |
Retrosynthesis Analysis
AI-Powered Synthesis Planning: Our tool employs the Template_relevance Pistachio, Template_relevance Bkms_metabolic, Template_relevance Pistachio_ringbreaker, Template_relevance Reaxys, Template_relevance Reaxys_biocatalysis model, leveraging a vast database of chemical reactions to predict feasible synthetic routes.
One-Step Synthesis Focus: Specifically designed for one-step synthesis, it provides concise and direct routes for your target compounds, streamlining the synthesis process.
Accurate Predictions: Utilizing the extensive PISTACHIO, BKMS_METABOLIC, PISTACHIO_RINGBREAKER, REAXYS, REAXYS_BIOCATALYSIS database, our tool offers high-accuracy predictions, reflecting the latest in chemical research and data.
Strategy Settings
| Precursor scoring | Relevance Heuristic |
|---|---|
| Min. plausibility | 0.01 |
| Model | Template_relevance |
| Template Set | Pistachio/Bkms_metabolic/Pistachio_ringbreaker/Reaxys/Reaxys_biocatalysis |
| Top-N result to add to graph | 6 |
Feasible Synthetic Routes
A: Tolazamide is a first-generation sulfonylurea drug that primarily acts on pancreatic β-cells by binding to sulfonylurea receptors (SUR1) on the cell membrane. [, , ] This binding blocks ATP-sensitive potassium channels, leading to membrane depolarization and the opening of voltage-gated calcium channels. [, , ] The influx of calcium ions triggers the exocytosis of insulin granules, increasing insulin secretion into the bloodstream. [, , ] This effectively lowers blood glucose levels, particularly in individuals with non-insulin-dependent diabetes mellitus (NIDDM), also known as type 2 diabetes. [, , ]
A:
- Molecular Formula: C14H21N3O3S []
- Molecular Weight: 311.4 g/mol []
- Spectroscopic Data: Key spectroscopic features include infrared (IR) and Raman spectra which provide information on functional group vibrations. [] Nuclear magnetic resonance (NMR) spectroscopy would be valuable to determine the structure and conformation. Single-crystal X-ray diffraction studies have been employed to fully elucidate the crystal structures of this compound polymorphs. []
A: this compound exists in different polymorphic forms, which can impact its stability and dissolution rate. [, ] Research suggests that specific amphiphilic block copolymers, like poly(ethylene glycol)-block-poly(lactic acid) (PEG-b-PLA), can act as crystal habit modifiers, influencing the shape of this compound crystals during crystallization. [] This modification may be beneficial in optimizing pharmaceutical formulations.
ANone: this compound is not typically recognized for its catalytic properties. Its primary mode of action revolves around binding to specific biological targets rather than catalyzing chemical reactions.
A: Yes, computational methods, including Density Functional Theory (DFT) calculations, have been used to investigate the potential of this compound and related sulfonylurea compounds as corrosion inhibitors for copper in nitric acid solutions. []
A: While specific SAR studies on this compound modifications are limited in the provided research, it is known that structural differences within the sulfonylurea class of drugs (including this compound) contribute to variations in their pharmacological properties such as duration of action, potency, and metabolism. []
A: this compound stability is influenced by factors such as pH and the presence of excipients. [] Research indicates that the block copolymer PEG-b-PLA can alter this compound's crystal habit, potentially influencing its dissolution rate and bioavailability. [] Formulation approaches would likely focus on controlling crystal form and particle size to optimize drug release and absorption.
ANone: As a pharmaceutical compound, this compound would be subject to stringent SHE regulations during its manufacturing, handling, and disposal. Specific regulations would vary based on geographical location and governing bodies.
A: this compound is well-absorbed after oral administration, with peak serum concentrations reached within 3-4 hours. [] It is extensively metabolized in the liver, primarily via oxidation and conjugation, with metabolites excreted in urine and feces. [] Its elimination half-life is approximately 7 hours. [] this compound's primary pharmacodynamic effect is the stimulation of insulin secretion from pancreatic β-cells, leading to a reduction in blood glucose levels. [, , ]
A: Numerous studies have explored the efficacy of this compound in the treatment of NIDDM. Animal studies using rat models have demonstrated that this compound potentiates insulin action in the liver and adipose tissue. [, , ] Clinical trials have shown its effectiveness in improving glycemic control in NIDDM patients, both as monotherapy and in combination with insulin. [, , , , , , , , ]
A: Secondary failure to sulfonylureas, including this compound, can occur in NIDDM patients, indicating a decline in drug effectiveness over time. [] This could be attributed to several factors, including a progressive decline in β-cell function and reduced insulin sensitivity. []
A: While generally considered safe for its intended use, this compound, like all medications, can cause side effects. [, , ] The most common adverse effects are hypoglycemia, gastrointestinal disturbances, and skin reactions. [, , ] In rare instances, more serious side effects like cholestatic jaundice and hepatic dysfunction have been reported. [, ]
A: Various analytical techniques have been employed to study this compound. High-pressure liquid chromatography (HPLC) is a common method for quantifying this compound levels in biological fluids. [] Gas chromatography (GC) has also been used to determine this compound concentrations in plasma. []
ANone: The provided research does not offer specific details on the environmental impact of this compound. As a pharmaceutical compound, its presence in the environment and potential effects would require further investigation.
A: Research has shown that this compound's dissolution rate varies significantly between different tablet formulations. [] This difference highlights the importance of considering formulation factors when assessing its bioavailability and efficacy.
A: Analytical methods for quantifying this compound, such as HPLC and GC, have established validation procedures. [, ] These procedures ensure the accuracy, precision, and specificity of the methods used for drug analysis.
ANone: As a pharmaceutical product, this compound production is subject to strict quality control and assurance measures throughout its lifecycle, from development and manufacturing to distribution. These measures ensure product consistency, safety, and efficacy.
ANone: The research provided does not focus on the immunogenicity of this compound. Further research would be needed to investigate any potential immune responses associated with its use.
ANone: The provided research does not specifically address drug-transporter interactions involving this compound. Further investigation would be required to understand the role of transporters in its pharmacokinetic profile.
ANone: The research provided does not directly address the biocompatibility or biodegradability of this compound. Further research would be needed to evaluate its long-term effects on biological systems and its environmental fate.
A: Several other sulfonylurea drugs, such as Glibenclamide and Glipizide, are available as alternatives to this compound. [] These drugs share a similar mechanism of action but may differ in their potency, duration of action, and side effect profiles. Biguanides, such as Metformin, represent another class of antidiabetic drugs with a distinct mechanism of action. [] The choice of medication depends on individual patient factors and should be determined by a healthcare professional.
ANone: Specific guidelines for the recycling and waste management of this compound would likely fall under pharmaceutical waste regulations, which vary depending on location. Proper disposal practices are crucial to minimize environmental contamination.
ANone: Research on this compound can benefit from various infrastructure and resources, including:
A: this compound, introduced in the 1960s, belongs to the first generation of sulfonylurea drugs developed for treating type 2 diabetes. [] Its introduction marked a significant advancement in diabetes management by providing an oral alternative to insulin injections. Research on this compound has contributed to a better understanding of pancreatic β-cell function, insulin secretion mechanisms, and the pathogenesis of type 2 diabetes.
Avertissement et informations sur les produits de recherche in vitro
Veuillez noter que tous les articles et informations sur les produits présentés sur BenchChem sont destinés uniquement à des fins informatives. Les produits disponibles à l'achat sur BenchChem sont spécifiquement conçus pour des études in vitro, qui sont réalisées en dehors des organismes vivants. Les études in vitro, dérivées du terme latin "in verre", impliquent des expériences réalisées dans des environnements de laboratoire contrôlés à l'aide de cellules ou de tissus. Il est important de noter que ces produits ne sont pas classés comme médicaments et n'ont pas reçu l'approbation de la FDA pour la prévention, le traitement ou la guérison de toute condition médicale, affection ou maladie. Nous devons souligner que toute forme d'introduction corporelle de ces produits chez les humains ou les animaux est strictement interdite par la loi. Il est essentiel de respecter ces directives pour assurer la conformité aux normes légales et éthiques en matière de recherche et d'expérimentation.