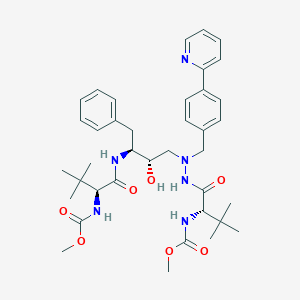
アタザナビル
概要
説明
Atazanavir is an antiretroviral medication used primarily to treat HIV/AIDS. It belongs to the protease inhibitor class and works by inhibiting the HIV protease enzyme, which is crucial for the maturation of infectious viral particles. Atazanavir is often used in combination with other antiretroviral agents to enhance its efficacy .
作用機序
Target of Action
Atazanavir, an antiviral protease inhibitor, primarily targets the human immunodeficiency virus type 1 (HIV-1) protease . This protease is a crucial enzyme that the HIV-1 virus requires for its multiplication .
Mode of Action
Atazanavir selectively inhibits the virus-specific processing of viral Gag and Gag-Pol polyproteins in HIV-1 infected cells . It achieves this by binding to the active site of the HIV-1 protease, thereby preventing the cleavage of the pro-form of viral proteins into the working machinery of the virus . This inhibition prevents the formation of mature virions, rendering the virus non-infectious .
Biochemical Pathways
The major biotransformation pathways of Atazanavir in humans consist of monooxygenation and dioxygenation . Other minor biotransformation pathways for Atazanavir or its metabolites consist of glucuronidation, N-dealkylation, hydrolysis, and oxygenation with dehydrogenation .
Pharmacokinetics
Atazanavir’s absorption, distribution, metabolism, and excretion (ADME) properties significantly impact its bioavailability. Atazanavir’s solubility decreases as pH increases, which can reduce plasma concentrations of Atazanavir if antacids, buffered medications, H2-receptor antagonists, and proton-pump inhibitors are administered with Atazanavir . Administration of Atazanavir with food enhances its bioavailability (35-70% increase in AUC) and reduces pharmacokinetic variability by 50% .
Result of Action
The molecular and cellular effects of Atazanavir’s action result in a decrease in the amount of HIV in the body . This, in turn, strengthens the immune system, reducing the risk of developing illnesses linked to HIV infection .
Action Environment
Environmental factors may influence the activity of Atazanavir, and drug-environment interactions may result in significantly altered absorption .
科学的研究の応用
Atazanavir has a wide range of scientific research applications:
Chemistry: It is used as a model compound in the study of protease inhibitors and their mechanisms.
Biology: Atazanavir is used in research to understand the lifecycle of HIV and the role of protease in viral maturation.
Industry: Atazanavir is used in the pharmaceutical industry for the development of antiretroviral therapies.
生化学分析
Biochemical Properties
Atazanavir interacts with the human immunodeficiency virus (HIV) protease, an enzyme crucial for the life cycle of the virus . The binding of Atazanavir to this enzyme inhibits its activity, preventing the maturation of viral particles .
Cellular Effects
Atazanavir influences cell function by inhibiting the HIV protease, thereby preventing the assembly and release of mature viral particles from infected cells . This results in a decrease in viral load and an increase in CD4 cell counts .
Molecular Mechanism
Atazanavir exerts its effects at the molecular level by binding to the active site of the HIV protease . This binding interaction inhibits the protease’s enzymatic activity, preventing the cleavage of viral polyproteins into individual functional proteins necessary for the assembly of mature viral particles .
Temporal Effects in Laboratory Settings
The effects of Atazanavir in laboratory settings have been observed to be stable over time . The emergence of Atazanavir-resistant strains of HIV, characterized by specific mutations in the viral protease, has been reported .
Dosage Effects in Animal Models
The effects of Atazanavir in animal models have not been explicitly mentioned in the sources. Like other antiretroviral drugs, the effectiveness of Atazanavir is likely to be dose-dependent, with higher doses resulting in greater inhibition of viral replication .
Metabolic Pathways
Atazanavir is involved in the metabolic pathway of HIV replication, where it interacts with the HIV protease . By inhibiting this enzyme, Atazanavir prevents the cleavage of viral polyproteins, a crucial step in the viral replication cycle .
Transport and Distribution
As a small molecule, Atazanavir is likely to be able to diffuse freely across cell membranes .
Subcellular Localization
Atazanavir, as a protease inhibitor, is likely to localize to the same subcellular compartments as the HIV protease . These are likely to be the cytoplasm and the endoplasmic reticulum, where viral polyprotein processing occurs .
準備方法
Synthetic Routes and Reaction Conditions
The synthesis of atazanavir involves multiple steps, including the use of condensing agents like DEPBT (Diethylphosphoryl cyanide) to facilitate the reaction between 1-[4-(pyridine-2-yl)-phenyl]-4(S)-hydroxy-5(S)-2,5-diamino-6-phenyl-2-azahexane and N-methoxycarbonyl-L-tertiary leucine in an organic solvent . This method is preferred due to its high yield and environmentally friendly nature.
Industrial Production Methods
In industrial settings, atazanavir is produced using a similar synthetic route but on a larger scale. The process involves the use of automated reactors and stringent quality control measures to ensure the purity and efficacy of the final product .
化学反応の分析
Types of Reactions
Atazanavir undergoes various chemical reactions, including:
Oxidation: Atazanavir can be oxidized to form hydroxylated and keto-metabolites.
Reduction: The compound can be reduced under specific conditions to yield different metabolites.
Substitution: Atazanavir can undergo substitution reactions, particularly in the presence of strong nucleophiles.
Common Reagents and Conditions
Oxidation: Common oxidizing agents include hydrogen peroxide and potassium permanganate.
Reduction: Reducing agents like sodium borohydride are often used.
Substitution: Strong nucleophiles such as sodium methoxide can facilitate substitution reactions.
Major Products Formed
The major products formed from these reactions include hydroxylated atazanavir, keto-atazanavir, and various substituted derivatives .
類似化合物との比較
Similar Compounds
Darunavir: Another protease inhibitor used in the treatment of HIV/AIDS.
Lopinavir: Often used in combination with ritonavir for enhanced efficacy.
Ritonavir: Used to boost the effectiveness of other protease inhibitors.
Uniqueness
Atazanavir is unique among protease inhibitors due to its once-daily dosing and lesser impact on lipid profiles, making it a preferred choice for patients with concerns about metabolic side effects . Additionally, atazanavir has shown effectiveness in patients with certain drug-resistant strains of HIV .
特性
IUPAC Name |
methyl N-[(2S)-1-[2-[(2S,3S)-2-hydroxy-3-[[(2S)-2-(methoxycarbonylamino)-3,3-dimethylbutanoyl]amino]-4-phenylbutyl]-2-[(4-pyridin-2-ylphenyl)methyl]hydrazinyl]-3,3-dimethyl-1-oxobutan-2-yl]carbamate | |
|---|---|---|
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
InChI |
InChI=1S/C38H52N6O7/c1-37(2,3)31(41-35(48)50-7)33(46)40-29(22-25-14-10-9-11-15-25)30(45)24-44(43-34(47)32(38(4,5)6)42-36(49)51-8)23-26-17-19-27(20-18-26)28-16-12-13-21-39-28/h9-21,29-32,45H,22-24H2,1-8H3,(H,40,46)(H,41,48)(H,42,49)(H,43,47)/t29-,30-,31+,32+/m0/s1 | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
InChI Key |
AXRYRYVKAWYZBR-GASGPIRDSA-N | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
Canonical SMILES |
CC(C)(C)C(C(=O)NC(CC1=CC=CC=C1)C(CN(CC2=CC=C(C=C2)C3=CC=CC=N3)NC(=O)C(C(C)(C)C)NC(=O)OC)O)NC(=O)OC | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
Isomeric SMILES |
CC(C)(C)[C@@H](C(=O)N[C@@H](CC1=CC=CC=C1)[C@H](CN(CC2=CC=C(C=C2)C3=CC=CC=N3)NC(=O)[C@H](C(C)(C)C)NC(=O)OC)O)NC(=O)OC | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
Molecular Formula |
C38H52N6O7 | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
DSSTOX Substance ID |
DTXSID9048691 | |
| Record name | Atazanavir | |
| Source | EPA DSSTox | |
| URL | https://comptox.epa.gov/dashboard/DTXSID9048691 | |
| Description | DSSTox provides a high quality public chemistry resource for supporting improved predictive toxicology. | |
Molecular Weight |
704.9 g/mol | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
Physical Description |
Solid | |
| Record name | Atazanavir | |
| Source | Human Metabolome Database (HMDB) | |
| URL | http://www.hmdb.ca/metabolites/HMDB0015205 | |
| Description | The Human Metabolome Database (HMDB) is a freely available electronic database containing detailed information about small molecule metabolites found in the human body. | |
| Explanation | HMDB is offered to the public as a freely available resource. Use and re-distribution of the data, in whole or in part, for commercial purposes requires explicit permission of the authors and explicit acknowledgment of the source material (HMDB) and the original publication (see the HMDB citing page). We ask that users who download significant portions of the database cite the HMDB paper in any resulting publications. | |
Solubility |
Free base slightly soluble (4-5 mg/mL), 3.27e-03 g/L | |
| Record name | Atazanavir | |
| Source | DrugBank | |
| URL | https://www.drugbank.ca/drugs/DB01072 | |
| Description | The DrugBank database is a unique bioinformatics and cheminformatics resource that combines detailed drug (i.e. chemical, pharmacological and pharmaceutical) data with comprehensive drug target (i.e. sequence, structure, and pathway) information. | |
| Explanation | Creative Common's Attribution-NonCommercial 4.0 International License (http://creativecommons.org/licenses/by-nc/4.0/legalcode) | |
| Record name | Atazanavir | |
| Source | Human Metabolome Database (HMDB) | |
| URL | http://www.hmdb.ca/metabolites/HMDB0015205 | |
| Description | The Human Metabolome Database (HMDB) is a freely available electronic database containing detailed information about small molecule metabolites found in the human body. | |
| Explanation | HMDB is offered to the public as a freely available resource. Use and re-distribution of the data, in whole or in part, for commercial purposes requires explicit permission of the authors and explicit acknowledgment of the source material (HMDB) and the original publication (see the HMDB citing page). We ask that users who download significant portions of the database cite the HMDB paper in any resulting publications. | |
Mechanism of Action |
Atazanavir selectively inhibits the virus-specific processing of viral Gag and Gag-Pol polyproteins in HIV-1 infected cells by binding to the active site of HIV-1 protease, thus preventing the formation of mature virions. Atazanavir is not active against HIV-2., Atazanavir is an azapeptide HIV-1 protease inhibitor. The compound selectively inhibits the virus-specific processing of viral Gag and Gag-Pol polyproteins in HIV-1 infected cells, thus preventing formation of mature virions., BMS-232632 is an azapeptide human immunodeficiency virus type 1 (HIV-1) protease (Prt) inhibitor that exhibits potent anti-HIV activity with a 50% effective concentration (EC(50)) of 2.6 to 5.3 nM and an EC(90) of 9 to 15 nM in cell culture. Proof-of-principle studies indicate that BMS-232632 blocks the cleavage of viral precursor proteins in HIV-infected cells, proving that it functions as an HIV Prt inhibitor. Comparative studies showed that BMS-232632 is generally more potent than the five currently approved HIV-1 Prt inhibitors. Furthermore, BMS-232632 is highly selective for HIV-1 Prt and exhibits cytotoxicity only at concentrations 6,500- to 23, 000-fold higher than that required for anti-HIV activity. To assess the potential of this inhibitor when used in combination with other antiretrovirals, BMS-232632 was evaluated for anti-HIV activity in two-drug combination studies. Combinations of BMS-232632 with either stavudine, didanosine, lamivudine, zidovudine, nelfinavir, indinavir, ritonavir, saquinavir, or amprenavir in HIV-infected peripheral blood mononuclear cells yielded additive to moderately synergistic antiviral effects. Importantly, combinations of drug pairs did not result in antagonistic anti-HIV activity or enhanced cytotoxic effects at the highest concentrations used for antiviral evaluation. Our results suggest that BMS-232632 may be an effective HIV-1 inhibitor that may be utilized in a variety of different drug combinations. | |
| Record name | Atazanavir | |
| Source | DrugBank | |
| URL | https://www.drugbank.ca/drugs/DB01072 | |
| Description | The DrugBank database is a unique bioinformatics and cheminformatics resource that combines detailed drug (i.e. chemical, pharmacological and pharmaceutical) data with comprehensive drug target (i.e. sequence, structure, and pathway) information. | |
| Explanation | Creative Common's Attribution-NonCommercial 4.0 International License (http://creativecommons.org/licenses/by-nc/4.0/legalcode) | |
| Record name | ATAZANAVIR | |
| Source | Hazardous Substances Data Bank (HSDB) | |
| URL | https://pubchem.ncbi.nlm.nih.gov/source/hsdb/7339 | |
| Description | The Hazardous Substances Data Bank (HSDB) is a toxicology database that focuses on the toxicology of potentially hazardous chemicals. It provides information on human exposure, industrial hygiene, emergency handling procedures, environmental fate, regulatory requirements, nanomaterials, and related areas. The information in HSDB has been assessed by a Scientific Review Panel. | |
CAS No. |
198904-31-3 | |
| Record name | Atazanavir | |
| Source | CAS Common Chemistry | |
| URL | https://commonchemistry.cas.org/detail?cas_rn=198904-31-3 | |
| Description | CAS Common Chemistry is an open community resource for accessing chemical information. Nearly 500,000 chemical substances from CAS REGISTRY cover areas of community interest, including common and frequently regulated chemicals, and those relevant to high school and undergraduate chemistry classes. This chemical information, curated by our expert scientists, is provided in alignment with our mission as a division of the American Chemical Society. | |
| Explanation | The data from CAS Common Chemistry is provided under a CC-BY-NC 4.0 license, unless otherwise stated. | |
| Record name | Atazanavir [INN:BAN] | |
| Source | ChemIDplus | |
| URL | https://pubchem.ncbi.nlm.nih.gov/substance/?source=chemidplus&sourceid=0198904313 | |
| Description | ChemIDplus is a free, web search system that provides access to the structure and nomenclature authority files used for the identification of chemical substances cited in National Library of Medicine (NLM) databases, including the TOXNET system. | |
| Record name | Atazanavir | |
| Source | DrugBank | |
| URL | https://www.drugbank.ca/drugs/DB01072 | |
| Description | The DrugBank database is a unique bioinformatics and cheminformatics resource that combines detailed drug (i.e. chemical, pharmacological and pharmaceutical) data with comprehensive drug target (i.e. sequence, structure, and pathway) information. | |
| Explanation | Creative Common's Attribution-NonCommercial 4.0 International License (http://creativecommons.org/licenses/by-nc/4.0/legalcode) | |
| Record name | Atazanavir | |
| Source | EPA DSSTox | |
| URL | https://comptox.epa.gov/dashboard/DTXSID9048691 | |
| Description | DSSTox provides a high quality public chemistry resource for supporting improved predictive toxicology. | |
| Record name | methyl N-[(2S)-1-[2-[(2S,3S)-2-hydroxy-3-[[(2S)-2-(methoxycarbonylamino)-3,3-dimethylbutanoyl]amino]-4-phenylbutyl]-2-[(4-pyridin-2-ylphenyl)methyl]hydrazinyl]-3,3-dimethyl-1-oxobutan-2-yl]carbamate | |
| Source | European Chemicals Agency (ECHA) | |
| URL | https://echa.europa.eu/substance-information/-/substanceinfo/100.243.594 | |
| Description | The European Chemicals Agency (ECHA) is an agency of the European Union which is the driving force among regulatory authorities in implementing the EU's groundbreaking chemicals legislation for the benefit of human health and the environment as well as for innovation and competitiveness. | |
| Explanation | Use of the information, documents and data from the ECHA website is subject to the terms and conditions of this Legal Notice, and subject to other binding limitations provided for under applicable law, the information, documents and data made available on the ECHA website may be reproduced, distributed and/or used, totally or in part, for non-commercial purposes provided that ECHA is acknowledged as the source: "Source: European Chemicals Agency, http://echa.europa.eu/". Such acknowledgement must be included in each copy of the material. ECHA permits and encourages organisations and individuals to create links to the ECHA website under the following cumulative conditions: Links can only be made to webpages that provide a link to the Legal Notice page. | |
| Record name | ATAZANAVIR | |
| Source | FDA Global Substance Registration System (GSRS) | |
| URL | https://gsrs.ncats.nih.gov/ginas/app/beta/substances/QZU4H47A3S | |
| Description | The FDA Global Substance Registration System (GSRS) enables the efficient and accurate exchange of information on what substances are in regulated products. Instead of relying on names, which vary across regulatory domains, countries, and regions, the GSRS knowledge base makes it possible for substances to be defined by standardized, scientific descriptions. | |
| Explanation | Unless otherwise noted, the contents of the FDA website (www.fda.gov), both text and graphics, are not copyrighted. They are in the public domain and may be republished, reprinted and otherwise used freely by anyone without the need to obtain permission from FDA. Credit to the U.S. Food and Drug Administration as the source is appreciated but not required. | |
| Record name | ATAZANAVIR | |
| Source | Hazardous Substances Data Bank (HSDB) | |
| URL | https://pubchem.ncbi.nlm.nih.gov/source/hsdb/7339 | |
| Description | The Hazardous Substances Data Bank (HSDB) is a toxicology database that focuses on the toxicology of potentially hazardous chemicals. It provides information on human exposure, industrial hygiene, emergency handling procedures, environmental fate, regulatory requirements, nanomaterials, and related areas. The information in HSDB has been assessed by a Scientific Review Panel. | |
| Record name | Atazanavir | |
| Source | Human Metabolome Database (HMDB) | |
| URL | http://www.hmdb.ca/metabolites/HMDB0015205 | |
| Description | The Human Metabolome Database (HMDB) is a freely available electronic database containing detailed information about small molecule metabolites found in the human body. | |
| Explanation | HMDB is offered to the public as a freely available resource. Use and re-distribution of the data, in whole or in part, for commercial purposes requires explicit permission of the authors and explicit acknowledgment of the source material (HMDB) and the original publication (see the HMDB citing page). We ask that users who download significant portions of the database cite the HMDB paper in any resulting publications. | |
Retrosynthesis Analysis
AI-Powered Synthesis Planning: Our tool employs the Template_relevance Pistachio, Template_relevance Bkms_metabolic, Template_relevance Pistachio_ringbreaker, Template_relevance Reaxys, Template_relevance Reaxys_biocatalysis model, leveraging a vast database of chemical reactions to predict feasible synthetic routes.
One-Step Synthesis Focus: Specifically designed for one-step synthesis, it provides concise and direct routes for your target compounds, streamlining the synthesis process.
Accurate Predictions: Utilizing the extensive PISTACHIO, BKMS_METABOLIC, PISTACHIO_RINGBREAKER, REAXYS, REAXYS_BIOCATALYSIS database, our tool offers high-accuracy predictions, reflecting the latest in chemical research and data.
Strategy Settings
| Precursor scoring | Relevance Heuristic |
|---|---|
| Min. plausibility | 0.01 |
| Model | Template_relevance |
| Template Set | Pistachio/Bkms_metabolic/Pistachio_ringbreaker/Reaxys/Reaxys_biocatalysis |
| Top-N result to add to graph | 6 |
Feasible Synthetic Routes
Q1: How does Atazanavir exert its anti-HIV effect?
A1: Atazanavir is a potent inhibitor of the HIV-1 aspartic protease enzyme. [, , , ] This enzyme is crucial for the cleavage of viral Gag and Gag-Pol polyproteins, a process essential for the formation of infectious HIV virions. [, ] By inhibiting this cleavage, Atazanavir prevents the maturation of new virus particles, effectively halting viral replication. [, ]
Q2: What is the chemical structure and molecular weight of Atazanavir?
A2: Atazanavir is an azapeptide with the chemical name (3S,8S,9S,12S)-3,12-bis(1,1-dimethylethyl)-8-hydroxy-4,11-dioxo-9-(phenylmethyl)-6-[[4-(2-pyridinyl)phenyl]methyl]-2,5,6,10,13-pentaazatetradecanedioic acid dimethyl ester, sulfate (1:1). [] Its molecular formula is C38H52N6O7•H2SO4, corresponding to a molecular weight of 802.9 g/mol (sulfuric acid salt). The free base has a molecular weight of 704.9 g/mol. []
Q3: Is spectroscopic data available for Atazanavir?
A3: While the provided research doesn't delve into detailed spectroscopic analysis, UV detection at 240 nm is a common method used in HPLC assays for quantifying Atazanavir. [, , ]
Q4: Which enzyme primarily metabolizes Atazanavir, and what are the implications for drug interactions?
A4: Atazanavir is primarily metabolized by the cytochrome P450 enzyme CYP3A4. [, , , ] This leads to significant drug interactions with other CYP3A4 inhibitors or inducers, requiring careful consideration during co-administration. [, , , ]
Q5: How does co-administration with Ritonavir affect Atazanavir pharmacokinetics?
A5: Ritonavir, a potent CYP3A4 inhibitor, is often co-administered with Atazanavir. [, , , , , , , , ] This co-administration "boosts" Atazanavir's plasma concentrations by inhibiting its metabolism, leading to increased drug exposure and allowing for once-daily dosing. [, , , , , , , , ]
Q6: How is Atazanavir absorbed and distributed in the body?
A6: Atazanavir exhibits pH-dependent solubility, impacting its absorption. [] Once absorbed, it demonstrates high protein binding in plasma, primarily to alpha-1 glycoprotein acid (AAG) and albumin. [, ] This protein binding influences its distribution and elimination. []
Q7: How do Atazanavir concentrations vary in different biological matrices?
A8: Atazanavir exhibits different concentrations across various biological matrices. [, ] Its concentrations in cerebrospinal fluid (CSF) are significantly lower than in plasma, potentially impacting its efficacy within the central nervous system. []
Q8: Do genetic factors influence Atazanavir pharmacokinetics?
A9: Yes, genetic polymorphisms in genes encoding drug-metabolizing enzymes and transporters impact Atazanavir pharmacokinetics. [, , ] For instance, individuals with the CYP3A5 expresser genotype exhibit faster Atazanavir clearance compared to non-expressers. [] Understanding these genetic influences paves the way for personalized dosing strategies.
Q9: What resistance patterns are associated with Atazanavir treatment failure?
A10: While Atazanavir displays a distinct resistance profile compared to other protease inhibitors, cross-resistance can occur in patients with prior protease inhibitor exposure. [, ] A unique I50L mutation in the HIV protease is frequently observed in treatment-naive patients experiencing virologic failure on Atazanavir-containing regimens. [, ] This mutation generally doesn't confer significant cross-resistance to other protease inhibitors. []
Q10: What strategies are employed to improve Atazanavir formulation and delivery?
A11: Research focuses on optimizing Atazanavir formulations to enhance its stability, solubility, and bioavailability. [] For example, the development of a bisulfate salt form aims to improve its crystalline properties and ultimately its pharmaceutical performance. []
Q11: What analytical techniques are commonly used to quantify Atazanavir?
A12: High-performance liquid chromatography (HPLC) coupled with various detection methods, such as UV detection and tandem mass spectrometry (MS/MS), are commonly employed to accurately quantify Atazanavir concentrations in biological samples. [, , , , ] These methods require rigorous validation to ensure accuracy, precision, and specificity for reliable research and clinical applications. [, ]
試験管内研究製品の免責事項と情報
BenchChemで提示されるすべての記事および製品情報は、情報提供を目的としています。BenchChemで購入可能な製品は、生体外研究のために特別に設計されています。生体外研究は、ラテン語の "in glass" に由来し、生物体の外で行われる実験を指します。これらの製品は医薬品または薬として分類されておらず、FDAから任何の医療状態、病気、または疾患の予防、治療、または治癒のために承認されていません。これらの製品を人間または動物に体内に導入する形態は、法律により厳格に禁止されています。これらのガイドラインに従うことは、研究と実験において法的および倫理的な基準の遵守を確実にするために重要です。


![3,3-Bis(trifluoromethyl)bicyclo[2.2.1]hept-5-ene-2,2-dicarbonitrile](/img/structure/B83.png)