リバスチグミン
概要
説明
Rivastigmine is a cholinesterase inhibitor used primarily for the treatment of mild to moderate dementia associated with Alzheimer’s disease and Parkinson’s disease . It works by inhibiting the breakdown of acetylcholine, a neurotransmitter crucial for memory and cognitive function . Rivastigmine is available in various forms, including oral capsules, liquid solutions, and transdermal patches .
科学的研究の応用
Rivastigmine has a wide range of scientific research applications:
作用機序
Target of Action
Rivastigmine primarily targets two enzymes in the brain: acetylcholinesterase and butyrylcholinesterase . These enzymes are responsible for breaking down a neurotransmitter called acetylcholine, which plays a crucial role in memory and cognition .
Mode of Action
Rivastigmine acts as a cholinesterase inhibitor . It binds reversibly with and inactivates cholinesterase enzymes, preventing the hydrolysis of acetylcholine . This action increases the concentration of acetylcholine at cholinergic synapses, enhancing cholinergic function .
Biochemical Pathways
The increased concentration of acetylcholine due to the inhibition of cholinesterase enzymes by Rivastigmine affects the cholinergic pathways in the brain . This is particularly relevant in conditions like Alzheimer’s disease, where there is a deficiency of acetylcholine due to the selective loss of cholinergic neurons in the cerebral cortex, nucleus basalis, and hippocampus .
Pharmacokinetics
Rivastigmine is well absorbed with a bioavailability of 60 to 72% . It crosses the blood-brain barrier, allowing it to act directly on the central nervous system . The major route of metabolism is through its target enzymes via cholinesterase-mediated hydrolysis .
Result of Action
The primary result of Rivastigmine’s action is an improvement in the symptoms of dementia, such as memory loss and cognitive deficits . This is achieved by enhancing the function of the cholinergic system through the increased concentration of acetylcholine . In addition, Rivastigmine has been shown to increase the levels of secreted amyloid precursor protein (sAPPα), which may have neuroprotective effects .
Action Environment
The efficacy and stability of Rivastigmine can be influenced by various environmental factors. For instance, the method of administration can affect its tolerability. Transdermal delivery of Rivastigmine, through a skin patch, has been shown to improve tolerability by lowering maximum plasma concentrations and prolonging time to maximum concentration .
生化学分析
Biochemical Properties
Rivastigmine, in addition to its anticholinesterase activity, directs APP processing away from BACE1 and towards α-secretases . It dose-dependently promotes α-secretase activity by upregulating levels of ADAM-9, -10, and -17 α-secretases in primary human brain cultures .
Cellular Effects
Rivastigmine has demonstrated treatment effects on the cognitive (thinking and memory), functional (activities of daily living), and behavioral problems commonly associated with Alzheimer’s . It improves cognition, participation in activities of daily living, and global evaluation ratings in patients with mild to moderately severe Alzheimer’s disease .
Molecular Mechanism
This is accomplished by increasing the concentration of acetylcholine through reversible inhibition of its hydrolysis by cholinesterase .
Temporal Effects in Laboratory Settings
Rivastigmine is rapidly metabolized by cholinesterase-mediated hydrolysis . The half-life in plasma is approximately 1 hour for oral Rivastigmine and 3.4 hours after transdermal patch .
Dosage Effects in Animal Models
Rivastigmine treatment elevated levels of sAPP in 3×Tg mice . Rivastigmine dose-dependently promoted α-secretase activity by upregulating levels of ADAM-9, -10, and -17 α-secretases in primary human brain cultures .
Metabolic Pathways
Rivastigmine is extensively metabolized primarily via cholinesterase-mediated hydrolysis to the decarbamylated metabolite NAP226-90 . Renal excretion of the metabolites is the major route of elimination .
Transport and Distribution
Thanks to its biochemical structure, Rivastigmine can cross the brain–blood barrier and acts mainly in the central nervous system . A minor proportion of Rivastigmine can also act at the neuromuscular junction, although its clinical effects are negligible at this level .
Subcellular Localization
Rivastigmine, being a cholinesterase inhibitor, is likely to be localized in the synaptic cleft where it prevents the breakdown of acetylcholine, thereby increasing the concentration of acetylcholine at cholinergic synapses .
準備方法
Synthetic Routes and Reaction Conditions: Rivastigmine can be synthesized through several methods. One common approach involves the esterification of m-hydroxyacetophenone, followed by asymmetric reductive amination, N-diphenylmethyl deprotection, and a final reductive amination step . The key transformation in this synthesis is the asymmetric reductive amination, which is catalyzed by an iridium–phosphoramidite ligand complex .
Industrial Production Methods: In industrial settings, rivastigmine is often produced using racemate resolution with tartaric acid derivatives . This method is favored due to its high yield and enantioselectivity. Additionally, transdermal patches of rivastigmine are developed using pressure-sensitive adhesives and controlled release membranes to ensure stable drug delivery over extended periods .
化学反応の分析
Types of Reactions: Rivastigmine undergoes various chemical reactions, including:
Oxidation: Rivastigmine can be oxidized under specific conditions, leading to the formation of its corresponding N-oxide derivatives.
Reduction: The compound can be reduced to its amine form using reducing agents like lithium aluminum hydride.
Substitution: Rivastigmine can participate in nucleophilic substitution reactions, where the carbamate group can be replaced by other nucleophiles.
Common Reagents and Conditions:
Oxidation: Hydrogen peroxide or m-chloroperbenzoic acid.
Reduction: Lithium aluminum hydride or sodium borohydride.
Substitution: Nucleophiles such as amines or thiols.
Major Products Formed:
Oxidation: N-oxide derivatives.
Reduction: Amine derivatives.
Substitution: Various substituted carbamates.
類似化合物との比較
Donepezil: Another cholinesterase inhibitor used for Alzheimer’s disease.
Galantamine: A cholinesterase inhibitor that also modulates nicotinic receptors.
Uniqueness of Rivastigmine: Rivastigmine’s ability to inhibit both acetylcholinesterase and butyrylcholinesterase sets it apart from other cholinesterase inhibitors . This dual inhibition may provide additional benefits in patients with more aggressive disease progression or those experiencing symptoms like hallucinations .
特性
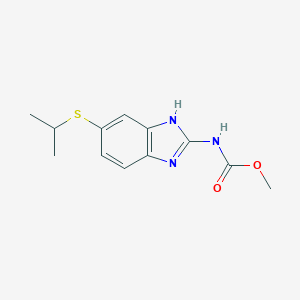
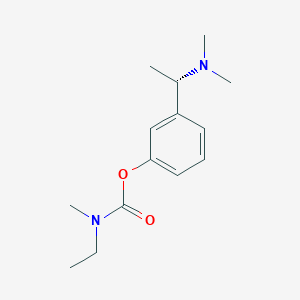
IUPAC Name |
[3-[(1S)-1-(dimethylamino)ethyl]phenyl] N-ethyl-N-methylcarbamate | |
|---|---|---|
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
InChI |
InChI=1S/C14H22N2O2/c1-6-16(5)14(17)18-13-9-7-8-12(10-13)11(2)15(3)4/h7-11H,6H2,1-5H3/t11-/m0/s1 | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
InChI Key |
XSVMFMHYUFZWBK-NSHDSACASA-N | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
Canonical SMILES |
CCN(C)C(=O)OC1=CC=CC(=C1)C(C)N(C)C | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
Isomeric SMILES |
CCN(C)C(=O)OC1=CC=CC(=C1)[C@H](C)N(C)C | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
Molecular Formula |
C14H22N2O2 | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
DSSTOX Substance ID |
DTXSID7023564 | |
| Record name | Rivastigmine | |
| Source | EPA DSSTox | |
| URL | https://comptox.epa.gov/dashboard/DTXSID7023564 | |
| Description | DSSTox provides a high quality public chemistry resource for supporting improved predictive toxicology. | |
Molecular Weight |
250.34 g/mol | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
Physical Description |
Solid | |
| Record name | Rivastigmine | |
| Source | Human Metabolome Database (HMDB) | |
| URL | http://www.hmdb.ca/metabolites/HMDB0015124 | |
| Description | The Human Metabolome Database (HMDB) is a freely available electronic database containing detailed information about small molecule metabolites found in the human body. | |
| Explanation | HMDB is offered to the public as a freely available resource. Use and re-distribution of the data, in whole or in part, for commercial purposes requires explicit permission of the authors and explicit acknowledgment of the source material (HMDB) and the original publication (see the HMDB citing page). We ask that users who download significant portions of the database cite the HMDB paper in any resulting publications. | |
Solubility |
2.04e+00 g/L | |
| Record name | Rivastigmine | |
| Source | Human Metabolome Database (HMDB) | |
| URL | http://www.hmdb.ca/metabolites/HMDB0015124 | |
| Description | The Human Metabolome Database (HMDB) is a freely available electronic database containing detailed information about small molecule metabolites found in the human body. | |
| Explanation | HMDB is offered to the public as a freely available resource. Use and re-distribution of the data, in whole or in part, for commercial purposes requires explicit permission of the authors and explicit acknowledgment of the source material (HMDB) and the original publication (see the HMDB citing page). We ask that users who download significant portions of the database cite the HMDB paper in any resulting publications. | |
Mechanism of Action |
Rivastigmine is a carbamate derivative that is structurally related to physostigmine, but not to donepezil and tacrine. The precise mechanism of rivastigmine has not been fully determined, but it is suggested that rivastigmine binds reversibly with and inactivates chlolinesterase (eg. acetylcholinesterase, butyrylcholinesterase), preventing the hydrolysis of acetycholine, and thus leading to an increased concentration of acetylcholine at cholinergic synapses. The anticholinesterase activity of rivastigmine is relatively specific for brain acetylcholinesterase and butyrylcholinesterase compared with those in peripheral tissues. | |
| Record name | Rivastigmine | |
| Source | DrugBank | |
| URL | https://www.drugbank.ca/drugs/DB00989 | |
| Description | The DrugBank database is a unique bioinformatics and cheminformatics resource that combines detailed drug (i.e. chemical, pharmacological and pharmaceutical) data with comprehensive drug target (i.e. sequence, structure, and pathway) information. | |
| Explanation | Creative Common's Attribution-NonCommercial 4.0 International License (http://creativecommons.org/licenses/by-nc/4.0/legalcode) | |
CAS No. |
123441-03-2 | |
| Record name | Rivastigmine | |
| Source | CAS Common Chemistry | |
| URL | https://commonchemistry.cas.org/detail?cas_rn=123441-03-2 | |
| Description | CAS Common Chemistry is an open community resource for accessing chemical information. Nearly 500,000 chemical substances from CAS REGISTRY cover areas of community interest, including common and frequently regulated chemicals, and those relevant to high school and undergraduate chemistry classes. This chemical information, curated by our expert scientists, is provided in alignment with our mission as a division of the American Chemical Society. | |
| Explanation | The data from CAS Common Chemistry is provided under a CC-BY-NC 4.0 license, unless otherwise stated. | |
| Record name | Rivastigmine [USAN:INN:BAN:JAN] | |
| Source | ChemIDplus | |
| URL | https://pubchem.ncbi.nlm.nih.gov/substance/?source=chemidplus&sourceid=0123441032 | |
| Description | ChemIDplus is a free, web search system that provides access to the structure and nomenclature authority files used for the identification of chemical substances cited in National Library of Medicine (NLM) databases, including the TOXNET system. | |
| Record name | Rivastigmine | |
| Source | DrugBank | |
| URL | https://www.drugbank.ca/drugs/DB00989 | |
| Description | The DrugBank database is a unique bioinformatics and cheminformatics resource that combines detailed drug (i.e. chemical, pharmacological and pharmaceutical) data with comprehensive drug target (i.e. sequence, structure, and pathway) information. | |
| Explanation | Creative Common's Attribution-NonCommercial 4.0 International License (http://creativecommons.org/licenses/by-nc/4.0/legalcode) | |
| Record name | Rivastigmine | |
| Source | EPA DSSTox | |
| URL | https://comptox.epa.gov/dashboard/DTXSID7023564 | |
| Description | DSSTox provides a high quality public chemistry resource for supporting improved predictive toxicology. | |
| Record name | Carbamic acid, N-ethyl-N-methyl-, 3-[(1S)-1-(dimethylamino)ethyl]phenyl ester | |
| Source | European Chemicals Agency (ECHA) | |
| URL | https://echa.europa.eu/substance-information/-/substanceinfo/100.120.679 | |
| Description | The European Chemicals Agency (ECHA) is an agency of the European Union which is the driving force among regulatory authorities in implementing the EU's groundbreaking chemicals legislation for the benefit of human health and the environment as well as for innovation and competitiveness. | |
| Explanation | Use of the information, documents and data from the ECHA website is subject to the terms and conditions of this Legal Notice, and subject to other binding limitations provided for under applicable law, the information, documents and data made available on the ECHA website may be reproduced, distributed and/or used, totally or in part, for non-commercial purposes provided that ECHA is acknowledged as the source: "Source: European Chemicals Agency, http://echa.europa.eu/". Such acknowledgement must be included in each copy of the material. ECHA permits and encourages organisations and individuals to create links to the ECHA website under the following cumulative conditions: Links can only be made to webpages that provide a link to the Legal Notice page. | |
| Record name | RIVASTIGMINE | |
| Source | FDA Global Substance Registration System (GSRS) | |
| URL | https://gsrs.ncats.nih.gov/ginas/app/beta/substances/PKI06M3IW0 | |
| Description | The FDA Global Substance Registration System (GSRS) enables the efficient and accurate exchange of information on what substances are in regulated products. Instead of relying on names, which vary across regulatory domains, countries, and regions, the GSRS knowledge base makes it possible for substances to be defined by standardized, scientific descriptions. | |
| Explanation | Unless otherwise noted, the contents of the FDA website (www.fda.gov), both text and graphics, are not copyrighted. They are in the public domain and may be republished, reprinted and otherwise used freely by anyone without the need to obtain permission from FDA. Credit to the U.S. Food and Drug Administration as the source is appreciated but not required. | |
| Record name | Rivastigmine | |
| Source | Human Metabolome Database (HMDB) | |
| URL | http://www.hmdb.ca/metabolites/HMDB0015124 | |
| Description | The Human Metabolome Database (HMDB) is a freely available electronic database containing detailed information about small molecule metabolites found in the human body. | |
| Explanation | HMDB is offered to the public as a freely available resource. Use and re-distribution of the data, in whole or in part, for commercial purposes requires explicit permission of the authors and explicit acknowledgment of the source material (HMDB) and the original publication (see the HMDB citing page). We ask that users who download significant portions of the database cite the HMDB paper in any resulting publications. | |
Synthesis routes and methods
Procedure details






Retrosynthesis Analysis
AI-Powered Synthesis Planning: Our tool employs the Template_relevance Pistachio, Template_relevance Bkms_metabolic, Template_relevance Pistachio_ringbreaker, Template_relevance Reaxys, Template_relevance Reaxys_biocatalysis model, leveraging a vast database of chemical reactions to predict feasible synthetic routes.
One-Step Synthesis Focus: Specifically designed for one-step synthesis, it provides concise and direct routes for your target compounds, streamlining the synthesis process.
Accurate Predictions: Utilizing the extensive PISTACHIO, BKMS_METABOLIC, PISTACHIO_RINGBREAKER, REAXYS, REAXYS_BIOCATALYSIS database, our tool offers high-accuracy predictions, reflecting the latest in chemical research and data.
Strategy Settings
| Precursor scoring | Relevance Heuristic |
|---|---|
| Min. plausibility | 0.01 |
| Model | Template_relevance |
| Template Set | Pistachio/Bkms_metabolic/Pistachio_ringbreaker/Reaxys/Reaxys_biocatalysis |
| Top-N result to add to graph | 6 |
Feasible Synthetic Routes
試験管内研究製品の免責事項と情報
BenchChemで提示されるすべての記事および製品情報は、情報提供を目的としています。BenchChemで購入可能な製品は、生体外研究のために特別に設計されています。生体外研究は、ラテン語の "in glass" に由来し、生物体の外で行われる実験を指します。これらの製品は医薬品または薬として分類されておらず、FDAから任何の医療状態、病気、または疾患の予防、治療、または治癒のために承認されていません。これらの製品を人間または動物に体内に導入する形態は、法律により厳格に禁止されています。これらのガイドラインに従うことは、研究と実験において法的および倫理的な基準の遵守を確実にするために重要です。