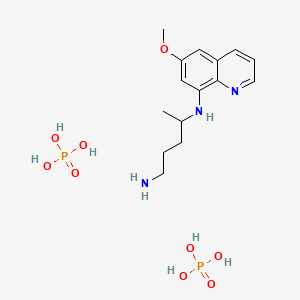
磷酸伯氨喹
描述
Primaquine phosphate is a synthetic compound with potent antimalarial activity. It is primarily used to treat and prevent malaria caused by Plasmodium vivax and Plasmodium ovale . Primaquine phosphate is an 8-aminoquinoline compound that eliminates tissue (exoerythrocytic) infection, thereby preventing the development of the blood (erythrocytic) forms of the parasite . It is also used to treat Pneumocystis pneumonia in combination with other medications .
科学研究应用
磷酸伯氨喹具有广泛的科学研究应用:
准备方法
合成路线和反应条件
磷酸伯氨喹是通过一系列化学反应从 8-氨基喹啉开始合成的。 反应条件通常涉及使用强碱和有机溶剂来促进取代反应 .
工业生产方法
磷酸伯氨喹的工业生产涉及使用优化反应条件的大规模合成,以确保高产率和纯度。 该过程包括通过结晶和过滤技术纯化最终产品 .
化学反应分析
反应类型
常用试剂和条件
形成的主要产物
相似化合物的比较
类似化合物
他弗诺喹: 另一种具有类似抗疟疾特性的 8-氨基喹啉类化合物.
氯喹: 一种用于治疗疟疾的 4-氨基喹啉类化合物.
甲氟喹: 一种用作抗疟疾的芳基氨基醇类化合物.
独特性
磷酸伯氨喹在消除疟原虫休眠的肝脏形式方面独一无二,可以防止复发 . 这种特性使其成为疟疾治疗方案的重要组成部分,特别是针对间日疟原虫和卵形疟原虫感染 .
属性
IUPAC Name |
4-N-(6-methoxyquinolin-8-yl)pentane-1,4-diamine;phosphoric acid | |
|---|---|---|
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
InChI |
InChI=1S/C15H21N3O.2H3O4P/c1-11(5-3-7-16)18-14-10-13(19-2)9-12-6-4-8-17-15(12)14;2*1-5(2,3)4/h4,6,8-11,18H,3,5,7,16H2,1-2H3;2*(H3,1,2,3,4) | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
InChI Key |
GJOHLWZHWQUKAU-UHFFFAOYSA-N | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
Canonical SMILES |
CC(CCCN)NC1=C2C(=CC(=C1)OC)C=CC=N2.OP(=O)(O)O.OP(=O)(O)O | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
Molecular Formula |
C15H27N3O9P2 | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
Related CAS |
90-34-6 (Parent) | |
| Record name | Primaquine phosphate [USP] | |
| Source | ChemIDplus | |
| URL | https://pubchem.ncbi.nlm.nih.gov/substance/?source=chemidplus&sourceid=0000063456 | |
| Description | ChemIDplus is a free, web search system that provides access to the structure and nomenclature authority files used for the identification of chemical substances cited in National Library of Medicine (NLM) databases, including the TOXNET system. | |
DSSTOX Substance ID |
DTXSID6045248 | |
| Record name | Primaquine diphosphate | |
| Source | EPA DSSTox | |
| URL | https://comptox.epa.gov/dashboard/DTXSID6045248 | |
| Description | DSSTox provides a high quality public chemistry resource for supporting improved predictive toxicology. | |
Molecular Weight |
455.34 g/mol | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
CAS No. |
63-45-6 | |
| Record name | Primaquine phosphate [USP] | |
| Source | ChemIDplus | |
| URL | https://pubchem.ncbi.nlm.nih.gov/substance/?source=chemidplus&sourceid=0000063456 | |
| Description | ChemIDplus is a free, web search system that provides access to the structure and nomenclature authority files used for the identification of chemical substances cited in National Library of Medicine (NLM) databases, including the TOXNET system. | |
| Record name | Primaquine diphosphate | |
| Source | EPA DSSTox | |
| URL | https://comptox.epa.gov/dashboard/DTXSID6045248 | |
| Description | DSSTox provides a high quality public chemistry resource for supporting improved predictive toxicology. | |
| Record name | Primaquine bis(phosphate) | |
| Source | European Chemicals Agency (ECHA) | |
| URL | https://echa.europa.eu/substance-information/-/substanceinfo/100.000.510 | |
| Description | The European Chemicals Agency (ECHA) is an agency of the European Union which is the driving force among regulatory authorities in implementing the EU's groundbreaking chemicals legislation for the benefit of human health and the environment as well as for innovation and competitiveness. | |
| Explanation | Use of the information, documents and data from the ECHA website is subject to the terms and conditions of this Legal Notice, and subject to other binding limitations provided for under applicable law, the information, documents and data made available on the ECHA website may be reproduced, distributed and/or used, totally or in part, for non-commercial purposes provided that ECHA is acknowledged as the source: "Source: European Chemicals Agency, http://echa.europa.eu/". Such acknowledgement must be included in each copy of the material. ECHA permits and encourages organisations and individuals to create links to the ECHA website under the following cumulative conditions: Links can only be made to webpages that provide a link to the Legal Notice page. | |
| Record name | PRIMAQUINE PHOSPHATE | |
| Source | FDA Global Substance Registration System (GSRS) | |
| URL | https://gsrs.ncats.nih.gov/ginas/app/beta/substances/H0982HF78B | |
| Description | The FDA Global Substance Registration System (GSRS) enables the efficient and accurate exchange of information on what substances are in regulated products. Instead of relying on names, which vary across regulatory domains, countries, and regions, the GSRS knowledge base makes it possible for substances to be defined by standardized, scientific descriptions. | |
| Explanation | Unless otherwise noted, the contents of the FDA website (www.fda.gov), both text and graphics, are not copyrighted. They are in the public domain and may be republished, reprinted and otherwise used freely by anyone without the need to obtain permission from FDA. Credit to the U.S. Food and Drug Administration as the source is appreciated but not required. | |
Retrosynthesis Analysis
AI-Powered Synthesis Planning: Our tool employs the Template_relevance Pistachio, Template_relevance Bkms_metabolic, Template_relevance Pistachio_ringbreaker, Template_relevance Reaxys, Template_relevance Reaxys_biocatalysis model, leveraging a vast database of chemical reactions to predict feasible synthetic routes.
One-Step Synthesis Focus: Specifically designed for one-step synthesis, it provides concise and direct routes for your target compounds, streamlining the synthesis process.
Accurate Predictions: Utilizing the extensive PISTACHIO, BKMS_METABOLIC, PISTACHIO_RINGBREAKER, REAXYS, REAXYS_BIOCATALYSIS database, our tool offers high-accuracy predictions, reflecting the latest in chemical research and data.
Strategy Settings
| Precursor scoring | Relevance Heuristic |
|---|---|
| Min. plausibility | 0.01 |
| Model | Template_relevance |
| Template Set | Pistachio/Bkms_metabolic/Pistachio_ringbreaker/Reaxys/Reaxys_biocatalysis |
| Top-N result to add to graph | 6 |
Feasible Synthetic Routes
Q1: What is the primary mechanism of action of Primaquine phosphate against malaria?
A1: While the exact mechanism remains unclear, Primaquine phosphate is known for its unique ability to target the dormant liver stage of Plasmodium vivax and Plasmodium ovale infections, called hypnozoites. [] This is crucial in preventing relapses, which are a significant challenge in treating these malaria species. []
Q2: How does Primaquine phosphate exert its anti-malarial effects?
A2: Several hypotheses exist regarding Primaquine phosphate's mechanism:
- Interference with Parasite Mitochondrial Function: It is suggested that Primaquine phosphate disrupts the electron transport chain in parasite mitochondria, ultimately leading to parasitic death. [, ]
- Formation of Reactive Oxygen Species: Primaquine phosphate may generate reactive oxygen species, causing oxidative damage within the parasite. []
- Inhibition of Parasite Nucleic Acid Synthesis: It has been proposed that Primaquine phosphate interferes with the synthesis of DNA and RNA in the parasite. []
Q3: What are the downstream effects of Primaquine phosphate on Plasmodium parasites?
A3: Primaquine phosphate's downstream effects are primarily linked to the proposed mechanisms mentioned above. These effects ultimately lead to the death of the parasite, effectively clearing the liver stage infection and preventing relapse.
Q4: What is the molecular formula and weight of Primaquine phosphate?
A4: The molecular formula of Primaquine phosphate is C15H21N3O·H3PO4. Its molecular weight is 365.3 g/mol.
Q5: Is there any spectroscopic data available for Primaquine phosphate?
A5: Yes, several studies have investigated Primaquine phosphate's spectroscopic properties: * FTIR spectroscopy: Used to identify characteristic functional groups and analyze potential interactions between Primaquine phosphate and excipients in formulations. [, ]* UV-Vis spectroscopy: Employed to assess the stability of Primaquine phosphate under various storage conditions. []
Q6: Does Primaquine phosphate exhibit any catalytic properties?
A6: While Primaquine phosphate is primarily recognized for its antimalarial activity, certain research explores its use as a spectrophotometric reagent for detecting trace amounts of gold. [] This application leverages Primaquine phosphate's ability to undergo oxidation by gold (III) ions in a sulfuric acid medium, forming a colored radical measurable at 550 nm.
Q7: Have there been any computational studies on Primaquine phosphate?
A7: While limited information is available on comprehensive computational modeling of Primaquine phosphate itself, some studies have focused on calculating physical parameters like molecular polarizability, diamagnetic susceptibility, and molecular electron ionization cross-section (Q). [] These parameters have been explored for potential correlations with drug activity, plasma protein binding, bioavailability, and dosage.
Q8: What formulation strategies have been investigated to enhance Primaquine phosphate's stability, solubility, or bioavailability?
A8: Several approaches have been explored to optimize Primaquine phosphate delivery:
- Solid Dispersions: Formulating Primaquine phosphate as a solid dispersion using carriers like mono ammonium glycyrrhizinate pentahydrate has been shown to effectively mask its bitter taste while also enhancing dissolution and potentially improving bioavailability. [, ]
- Nanoparticles: Encapsulating Primaquine phosphate within nanoparticles, such as PLGA nanoparticles and PEGylated galactosylated nano lipid carriers, has shown promise in enhancing drug loading, prolonging drug release, and potentially improving its targeting to the liver. [, ] These strategies aim to improve the drug's therapeutic efficacy and minimize potential side effects.
Q9: What are the key considerations for Safety, Health, and Environment (SHE) regulations concerning Primaquine phosphate?
A9: While the provided excerpts do not explicitly detail SHE regulations, responsible handling and disposal of Primaquine phosphate are crucial due to its potential environmental impacts.
Q10: What is the pharmacokinetic profile of Primaquine phosphate?
A10: Primaquine phosphate exhibits a relatively short half-life, necessitating frequent administration and potentially contributing to adverse effects and patient non-compliance. [] Various studies highlighted the pharmacokinetic properties of Primaquine phosphate:
- Absorption and Metabolism: Primaquine phosphate is well-absorbed after oral administration, and its metabolism involves the cytochrome P450 enzyme, particularly CYP2D6. []
- Bioavailability and Distribution: Studies have explored methods to improve Primaquine phosphate's bioavailability and target its delivery to the liver, the primary site of hypnozoite residence. []
- Excretion: The drug and its metabolites are primarily excreted in the urine. []
Q11: What is the relationship between Primaquine phosphate's pharmacokinetics and its pharmacodynamics?
A11: Research indicates a link between impaired CYP2D6 activity and decreased efficacy of Primaquine phosphate in achieving radical cure for P. vivax malaria. [] Individuals with compromised CYP2D6 activity, either due to genetic variations or drug interactions, may experience a higher risk of relapse.
Q12: What evidence supports the in vitro and in vivo efficacy of Primaquine phosphate?
A12: * In vitro: Studies demonstrate Primaquine phosphate's efficacy against Toxoplasma gondii tachyzoites in vitro, inhibiting their growth at specific concentrations. []* In vivo: In animal models, specifically Plasmodium berghei-infected mice, Primaquine phosphate formulated as solid lipid nanoparticles exhibited enhanced effectiveness compared to conventional oral doses. [] This highlights the potential of nanoformulations in improving the drug's therapeutic index.
Q13: What are the known mechanisms of resistance to Primaquine phosphate?
A13: While the precise mechanisms of resistance to Primaquine phosphate are not fully understood, they are thought to involve alterations in parasite drug uptake, drug target modifications, or enhanced detoxification pathways. []
Q14: Is there evidence of cross-resistance between Primaquine phosphate and other antimalarials?
A14: Although the provided excerpts do not directly address cross-resistance, it's a concern with antimalarial drugs. The emergence of resistance to one antimalarial can potentially reduce the efficacy of others with similar mechanisms of action.
Q15: What are the known toxicological effects and safety concerns associated with Primaquine phosphate?
A15: Primaquine phosphate can cause hemolytic anemia, particularly in individuals with glucose-6-phosphate dehydrogenase (G6PD) deficiency. [, , , ] Screening for G6PD deficiency before Primaquine phosphate administration is crucial to prevent this serious adverse effect. []
Q16: What drug delivery strategies have been explored to improve Primaquine phosphate's targeting to the liver?
A16: Researchers are investigating nanocarriers, like galactose-anchored gelatin nanoparticles, to specifically deliver Primaquine phosphate to the liver. [] This approach exploits the asialoglycoprotein receptor expressed on hepatocytes for targeted drug delivery.
Q17: Are there any biomarkers associated with Primaquine phosphate efficacy or toxicity?
A17: CYP2D6 genotype and phenotype can serve as biomarkers to predict the efficacy of Primaquine phosphate. Impaired CYP2D6 activity is linked to a higher relapse risk, indicating potential treatment failure. [] G6PD deficiency is a crucial biomarker for Primaquine phosphate-induced hemolytic anemia. [, , , ]
Q18: What analytical techniques are commonly employed in Primaquine phosphate research?
A18: Several analytical methods are crucial in Primaquine phosphate research:* High-Performance Liquid Chromatography (HPLC): A primary technique for quantitative determination of Primaquine phosphate in various matrices, including tablets, biological samples, and formulations. [, , , ]* Spectrophotometry: Employed to determine Primaquine phosphate concentrations by measuring the absorbance of colored complexes formed with specific reagents. [, , , ]* Dissolution Testing: Used to evaluate the release rate of Primaquine phosphate from various formulations, such as tablets, and assess how different factors impact its dissolution profile. []
Q19: Are there any viable alternatives or substitutes for Primaquine phosphate in treating malaria?
A19: While Primaquine phosphate remains a crucial drug for preventing relapse in specific malaria species, researchers are exploring alternative therapies: * Tafenoquine: A single-dose 8-aminoquinoline antimalarial drug with a longer half-life than Primaquine phosphate, potentially improving patient adherence. []
体外研究产品的免责声明和信息
请注意,BenchChem 上展示的所有文章和产品信息仅供信息参考。 BenchChem 上可购买的产品专为体外研究设计,这些研究在生物体外进行。体外研究,源自拉丁语 "in glass",涉及在受控实验室环境中使用细胞或组织进行的实验。重要的是要注意,这些产品没有被归类为药物或药品,他们没有得到 FDA 的批准,用于预防、治疗或治愈任何医疗状况、疾病或疾病。我们必须强调,将这些产品以任何形式引入人类或动物的身体都是法律严格禁止的。遵守这些指南对确保研究和实验的法律和道德标准的符合性至关重要。