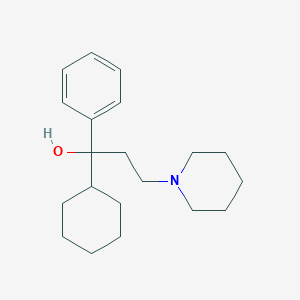
Trihexyphenidyl
概要
説明
トリヘキシフェニジルは、主にパーキンソン病の症状や薬物誘発性錐体外路症状の管理に使用される合成抗痙攣薬です。 これは抗コリン薬に分類され、こわばり、振戦、痙攣、筋力低下などの症状を軽減する効果で知られています 。 トリヘキシフェニジルは1949年にFDAによって初めて承認され、それ以来、さまざまな形態のパーキンソン症の症状治療において貴重なツールとなっています .
2. 製法
トリヘキシフェニジルの合成には、いくつかの重要なステップが含まれます。
中間体プロピオフェノン・ピペリジル塩酸塩の合成: この中間体は、マンニッヒ反応を用いて、アセトフェノンをパラホルムアルデヒドとピペリジンでアミノメチル化することにより調製されます.
粗トリヘキシフェニジル塩酸塩の生成: 次に、この中間体をグリニャール試薬と反応させます。通常、ジエチルエーテルに関連する安全上の問題を回避するために、メチルターシャリーブチルエーテルを溶媒として使用します.
準備方法
The synthesis of trihexyphenidyl involves several key steps:
Synthesis of Intermediate Propiophenone Piperidyl Hydrochloride: This intermediate is prepared through the aminomethylation of acetophenone using paraformaldehyde and piperidine in a Mannich reaction.
Formation of Crude this compound Hydrochloride: The intermediate is then reacted with a Grignard reagent, typically using methyl tertiary butyl ether as a solvent to avoid the safety issues associated with diethyl ether.
Refinement: The crude product is refined to obtain pure this compound hydrochloride.
化学反応の分析
トリヘキシフェニジルは、次のようなさまざまな化学反応を起こします。
酸化: 特定の条件下で酸化される可能性がありますが、これは治療用途では一般的ではありません。
還元: 還元反応はそれほど一般的ではありませんが、制御された実験室条件下で実行できます。
これらの反応に使用される一般的な試薬には、過マンガン酸カリウムなどの酸化剤と、水素化リチウムアルミニウムなどの還元剤が含まれます。生成される主な生成物は、使用する特定の反応条件と試薬によって異なります。
科学的研究の応用
トリヘキシフェニジルは、幅広い科学研究に応用されています。
作用機序
トリヘキシフェニジルは、M1サブタイプに対して高い親和性を持ち、非選択的なムスカリン性アセチルコリン受容体拮抗薬として作用することにより効果を発揮します 。副交感神経を阻害することにより、筋肉の硬直と振戦を軽減します。 この薬物は、平滑筋に直接的な痙攣解作用を有し、弱い散瞳作用、唾液分泌抑制作用、心臓迷走神経阻害作用を示します .
類似化合物との比較
トリヘキシフェニジルは、しばしば次のような他の抗コリン薬と比較されます。
ベンztropine: 抗コリン作用は似ていますが、薬物動態特性が異なります.
ジフェンヒドラミン: 抗ヒスタミン剤であり、抗コリン作用を有し、同様の適応症で使用されますが、より幅広い用途があります.
トリヘキシフェニジルは、中枢ムスカリン受容体に対する特異的な親和性を持つため、中枢神経系に関連する症状の管理に特に効果的です .
特性
IUPAC Name |
1-cyclohexyl-1-phenyl-3-piperidin-1-ylpropan-1-ol | |
|---|---|---|
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
InChI |
InChI=1S/C20H31NO/c22-20(18-10-4-1-5-11-18,19-12-6-2-7-13-19)14-17-21-15-8-3-9-16-21/h1,4-5,10-11,19,22H,2-3,6-9,12-17H2 | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
InChI Key |
HWHLPVGTWGOCJO-UHFFFAOYSA-N | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
Canonical SMILES |
C1CCC(CC1)C(CCN2CCCCC2)(C3=CC=CC=C3)O | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
Molecular Formula |
C20H31NO | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
Related CAS |
52-49-3 (hydrochloride) | |
| Record name | Trihexyphenidyl [INN:BAN] | |
| Source | ChemIDplus | |
| URL | https://pubchem.ncbi.nlm.nih.gov/substance/?source=chemidplus&sourceid=0000144116 | |
| Description | ChemIDplus is a free, web search system that provides access to the structure and nomenclature authority files used for the identification of chemical substances cited in National Library of Medicine (NLM) databases, including the TOXNET system. | |
DSSTOX Substance ID |
DTXSID4023705 | |
| Record name | Trihexyphenidyl | |
| Source | EPA DSSTox | |
| URL | https://comptox.epa.gov/dashboard/DTXSID4023705 | |
| Description | DSSTox provides a high quality public chemistry resource for supporting improved predictive toxicology. | |
Molecular Weight |
301.5 g/mol | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
Physical Description |
Solid | |
| Record name | Trihexyphenidyl | |
| Source | Human Metabolome Database (HMDB) | |
| URL | http://www.hmdb.ca/metabolites/HMDB0014520 | |
| Description | The Human Metabolome Database (HMDB) is a freely available electronic database containing detailed information about small molecule metabolites found in the human body. | |
| Explanation | HMDB is offered to the public as a freely available resource. Use and re-distribution of the data, in whole or in part, for commercial purposes requires explicit permission of the authors and explicit acknowledgment of the source material (HMDB) and the original publication (see the HMDB citing page). We ask that users who download significant portions of the database cite the HMDB paper in any resulting publications. | |
Solubility |
Crystals; decomp at 258.5 °C; pH of 1% aq soln 5.5-6.0; solubility (g/100 mL): water at 25 °C 1.0, alcohol 6, chloroform 5; more soluble in methanol; very slightly soluble in ether, benzene /Hydrochloride/, 3.14e-03 g/L | |
| Record name | TRIHEXYPHENIDYL | |
| Source | Hazardous Substances Data Bank (HSDB) | |
| URL | https://pubchem.ncbi.nlm.nih.gov/source/hsdb/3196 | |
| Description | The Hazardous Substances Data Bank (HSDB) is a toxicology database that focuses on the toxicology of potentially hazardous chemicals. It provides information on human exposure, industrial hygiene, emergency handling procedures, environmental fate, regulatory requirements, nanomaterials, and related areas. The information in HSDB has been assessed by a Scientific Review Panel. | |
| Record name | Trihexyphenidyl | |
| Source | Human Metabolome Database (HMDB) | |
| URL | http://www.hmdb.ca/metabolites/HMDB0014520 | |
| Description | The Human Metabolome Database (HMDB) is a freely available electronic database containing detailed information about small molecule metabolites found in the human body. | |
| Explanation | HMDB is offered to the public as a freely available resource. Use and re-distribution of the data, in whole or in part, for commercial purposes requires explicit permission of the authors and explicit acknowledgment of the source material (HMDB) and the original publication (see the HMDB citing page). We ask that users who download significant portions of the database cite the HMDB paper in any resulting publications. | |
Mechanism of Action |
Trihexyphenidyl is a non-selective muscarinic acetylcholine receptor antagonist but binds with higher affinity to the M1 subtype. In vivo studies have shown that trihexyphenidyl demonstrates higher affinity for central muscarinic receptors located in the cerebral cortex and lower affinity for those located peripherally. Other studies suggest that trihexyphenidyl may modify nicotinic acetylcholine receptor neurotransmission, leading indirectly to enhanced dopamine release in the striatum. Although the anticholinergic has proven to be useful in the treatment of symptoms associated with Parkinson’s disease or other movement disorders, its mechanism of action has yet to be fully elucidated., Cerebral blood flow and oxygen metabolism were studied in six previously untreated patients with Parkinson's disease (PD) before and after anticholinergic treatment using positron emission tomography (PET) and compared with six controls. The PET study and an assessment of the disability and cognitive impairment were performed before and after administration of 6 mg trihexyphenidyl for 5 to 11 weeks. All PD patients showed improvements in motor symptoms after the trihexyphenidyl treatment. Cognitive function did not significantly differ between before and after trihexyphenidyl treatment. However, after trihexyphenidyl treatment, regional cerebral blood flow (rCBF) and regional oxygen metabolic rate (rCMRO2) decreased by 15% in the striatum and by 10% in all cortical areas contralateral to predominantly symptomatic limbs, and by 10% in the ipsilateral striatum and all cortical areas, significantly below the values of controls in most cerebral cortices and striatum. These findings suggest that trihexyphenidyl inhibits the cortical cholinergic system and significantly decreases rCBF and rCMRO2 in the cerebral cortices without cognitive impairment in untreated patients with PD., In common with other antimuscarinic agents, trihexyphenidyl produces an atropine-like blocking action on parasympathetic-innervated peripheral structures, including smooth muscle. In addition, trihexyphenidyl exhibits a direct spasmolytic action on smooth muscle and exhibits weak mydriatic, antisialagogue, and cardiovagal blocking effects. The exact mechanism of action of trihexyphenidyl in parkinsonian syndrome is not understood but may result from blockade of efferent impulses and from central inhibition of cerebral motor centers. In small doses, trihexyphenidyl depresses the CNS but larger doses cause cerebral excitement resembling the signs of atropine toxicity., In vivo microdialysis was used to study the effect of the non-selective muscarinic antagonist, trihexyphenidyl, on the decarboxylation of levodopa (L-dopa) in the striatum of hemi-Parkinson rats. In normal rats, continuous perfusion of trihexyphenidyl (1 mM) via the microdialysis probe induced a significant increase in striatal dopamine release, followed by a decrease to below baseline values. A similar effect was observed, though less pronounced, in denervated striatum of rats with a unilateral 6-hydroxydopamine lesion of the nigrostriatal pathway. In these hemi-Parkinson rats, continuous striatal perfusion of trihexyphenidyl had no effect on the biotransformation of locally applied L-dopa (2 uM for 20 min) to dopamine in either intact or denervated striatum. However, systemic administration of trihexyphenidyl (1.5 mg/kg ip) produced an attenuation of the L- dopa-induced dopamine release in the intact striatum (contralateral to the lesion) of hemi-Parkinson rats. This effect was absent in the denervated striatum of these animals. We confirmed that L-dopa induces an increase in striatal dopamine output which is influenced by the severity of the dopaminergic denervation. The absence of an effect of trihexyphenidyl locally applied in the striatum, on biotransformation of L-dopa suggests that the site of action of antimuscarinic drugs may not be in the striatum and, therefore, remains unclear. | |
| Record name | Trihexyphenidyl | |
| Source | DrugBank | |
| URL | https://www.drugbank.ca/drugs/DB00376 | |
| Description | The DrugBank database is a unique bioinformatics and cheminformatics resource that combines detailed drug (i.e. chemical, pharmacological and pharmaceutical) data with comprehensive drug target (i.e. sequence, structure, and pathway) information. | |
| Explanation | Creative Common's Attribution-NonCommercial 4.0 International License (http://creativecommons.org/licenses/by-nc/4.0/legalcode) | |
| Record name | TRIHEXYPHENIDYL | |
| Source | Hazardous Substances Data Bank (HSDB) | |
| URL | https://pubchem.ncbi.nlm.nih.gov/source/hsdb/3196 | |
| Description | The Hazardous Substances Data Bank (HSDB) is a toxicology database that focuses on the toxicology of potentially hazardous chemicals. It provides information on human exposure, industrial hygiene, emergency handling procedures, environmental fate, regulatory requirements, nanomaterials, and related areas. The information in HSDB has been assessed by a Scientific Review Panel. | |
Impurities |
1-phenyl-3-(piperidine-1-yl)propan-1-one | |
| Record name | TRIHEXYPHENIDYL | |
| Source | Hazardous Substances Data Bank (HSDB) | |
| URL | https://pubchem.ncbi.nlm.nih.gov/source/hsdb/3196 | |
| Description | The Hazardous Substances Data Bank (HSDB) is a toxicology database that focuses on the toxicology of potentially hazardous chemicals. It provides information on human exposure, industrial hygiene, emergency handling procedures, environmental fate, regulatory requirements, nanomaterials, and related areas. The information in HSDB has been assessed by a Scientific Review Panel. | |
CAS No. |
144-11-6 | |
| Record name | (±)-Trihexyphenidyl | |
| Source | CAS Common Chemistry | |
| URL | https://commonchemistry.cas.org/detail?cas_rn=144-11-6 | |
| Description | CAS Common Chemistry is an open community resource for accessing chemical information. Nearly 500,000 chemical substances from CAS REGISTRY cover areas of community interest, including common and frequently regulated chemicals, and those relevant to high school and undergraduate chemistry classes. This chemical information, curated by our expert scientists, is provided in alignment with our mission as a division of the American Chemical Society. | |
| Explanation | The data from CAS Common Chemistry is provided under a CC-BY-NC 4.0 license, unless otherwise stated. | |
| Record name | Trihexyphenidyl [INN:BAN] | |
| Source | ChemIDplus | |
| URL | https://pubchem.ncbi.nlm.nih.gov/substance/?source=chemidplus&sourceid=0000144116 | |
| Description | ChemIDplus is a free, web search system that provides access to the structure and nomenclature authority files used for the identification of chemical substances cited in National Library of Medicine (NLM) databases, including the TOXNET system. | |
| Record name | Trihexyphenidyl | |
| Source | DrugBank | |
| URL | https://www.drugbank.ca/drugs/DB00376 | |
| Description | The DrugBank database is a unique bioinformatics and cheminformatics resource that combines detailed drug (i.e. chemical, pharmacological and pharmaceutical) data with comprehensive drug target (i.e. sequence, structure, and pathway) information. | |
| Explanation | Creative Common's Attribution-NonCommercial 4.0 International License (http://creativecommons.org/licenses/by-nc/4.0/legalcode) | |
| Record name | trihexyphenidyl | |
| Source | DTP/NCI | |
| URL | https://dtp.cancer.gov/dtpstandard/servlet/dwindex?searchtype=NSC&outputformat=html&searchlist=12268 | |
| Description | The NCI Development Therapeutics Program (DTP) provides services and resources to the academic and private-sector research communities worldwide to facilitate the discovery and development of new cancer therapeutic agents. | |
| Explanation | Unless otherwise indicated, all text within NCI products is free of copyright and may be reused without our permission. Credit the National Cancer Institute as the source. | |
| Record name | Trihexyphenidyl | |
| Source | EPA DSSTox | |
| URL | https://comptox.epa.gov/dashboard/DTXSID4023705 | |
| Description | DSSTox provides a high quality public chemistry resource for supporting improved predictive toxicology. | |
| Record name | Trihexyphenidyl | |
| Source | European Chemicals Agency (ECHA) | |
| URL | https://echa.europa.eu/substance-information/-/substanceinfo/100.005.105 | |
| Description | The European Chemicals Agency (ECHA) is an agency of the European Union which is the driving force among regulatory authorities in implementing the EU's groundbreaking chemicals legislation for the benefit of human health and the environment as well as for innovation and competitiveness. | |
| Explanation | Use of the information, documents and data from the ECHA website is subject to the terms and conditions of this Legal Notice, and subject to other binding limitations provided for under applicable law, the information, documents and data made available on the ECHA website may be reproduced, distributed and/or used, totally or in part, for non-commercial purposes provided that ECHA is acknowledged as the source: "Source: European Chemicals Agency, http://echa.europa.eu/". Such acknowledgement must be included in each copy of the material. ECHA permits and encourages organisations and individuals to create links to the ECHA website under the following cumulative conditions: Links can only be made to webpages that provide a link to the Legal Notice page. | |
| Record name | TRIHEXYPHENIDYL | |
| Source | FDA Global Substance Registration System (GSRS) | |
| URL | https://gsrs.ncats.nih.gov/ginas/app/beta/substances/6RC5V8B7PO | |
| Description | The FDA Global Substance Registration System (GSRS) enables the efficient and accurate exchange of information on what substances are in regulated products. Instead of relying on names, which vary across regulatory domains, countries, and regions, the GSRS knowledge base makes it possible for substances to be defined by standardized, scientific descriptions. | |
| Explanation | Unless otherwise noted, the contents of the FDA website (www.fda.gov), both text and graphics, are not copyrighted. They are in the public domain and may be republished, reprinted and otherwise used freely by anyone without the need to obtain permission from FDA. Credit to the U.S. Food and Drug Administration as the source is appreciated but not required. | |
| Record name | TRIHEXYPHENIDYL | |
| Source | Hazardous Substances Data Bank (HSDB) | |
| URL | https://pubchem.ncbi.nlm.nih.gov/source/hsdb/3196 | |
| Description | The Hazardous Substances Data Bank (HSDB) is a toxicology database that focuses on the toxicology of potentially hazardous chemicals. It provides information on human exposure, industrial hygiene, emergency handling procedures, environmental fate, regulatory requirements, nanomaterials, and related areas. The information in HSDB has been assessed by a Scientific Review Panel. | |
| Record name | Trihexyphenidyl | |
| Source | Human Metabolome Database (HMDB) | |
| URL | http://www.hmdb.ca/metabolites/HMDB0014520 | |
| Description | The Human Metabolome Database (HMDB) is a freely available electronic database containing detailed information about small molecule metabolites found in the human body. | |
| Explanation | HMDB is offered to the public as a freely available resource. Use and re-distribution of the data, in whole or in part, for commercial purposes requires explicit permission of the authors and explicit acknowledgment of the source material (HMDB) and the original publication (see the HMDB citing page). We ask that users who download significant portions of the database cite the HMDB paper in any resulting publications. | |
Melting Point |
114 °C, 258.5 °C | |
| Record name | Trihexyphenidyl | |
| Source | DrugBank | |
| URL | https://www.drugbank.ca/drugs/DB00376 | |
| Description | The DrugBank database is a unique bioinformatics and cheminformatics resource that combines detailed drug (i.e. chemical, pharmacological and pharmaceutical) data with comprehensive drug target (i.e. sequence, structure, and pathway) information. | |
| Explanation | Creative Common's Attribution-NonCommercial 4.0 International License (http://creativecommons.org/licenses/by-nc/4.0/legalcode) | |
| Record name | TRIHEXYPHENIDYL | |
| Source | Hazardous Substances Data Bank (HSDB) | |
| URL | https://pubchem.ncbi.nlm.nih.gov/source/hsdb/3196 | |
| Description | The Hazardous Substances Data Bank (HSDB) is a toxicology database that focuses on the toxicology of potentially hazardous chemicals. It provides information on human exposure, industrial hygiene, emergency handling procedures, environmental fate, regulatory requirements, nanomaterials, and related areas. The information in HSDB has been assessed by a Scientific Review Panel. | |
| Record name | Trihexyphenidyl | |
| Source | Human Metabolome Database (HMDB) | |
| URL | http://www.hmdb.ca/metabolites/HMDB0014520 | |
| Description | The Human Metabolome Database (HMDB) is a freely available electronic database containing detailed information about small molecule metabolites found in the human body. | |
| Explanation | HMDB is offered to the public as a freely available resource. Use and re-distribution of the data, in whole or in part, for commercial purposes requires explicit permission of the authors and explicit acknowledgment of the source material (HMDB) and the original publication (see the HMDB citing page). We ask that users who download significant portions of the database cite the HMDB paper in any resulting publications. | |
Retrosynthesis Analysis
AI-Powered Synthesis Planning: Our tool employs the Template_relevance Pistachio, Template_relevance Bkms_metabolic, Template_relevance Pistachio_ringbreaker, Template_relevance Reaxys, Template_relevance Reaxys_biocatalysis model, leveraging a vast database of chemical reactions to predict feasible synthetic routes.
One-Step Synthesis Focus: Specifically designed for one-step synthesis, it provides concise and direct routes for your target compounds, streamlining the synthesis process.
Accurate Predictions: Utilizing the extensive PISTACHIO, BKMS_METABOLIC, PISTACHIO_RINGBREAKER, REAXYS, REAXYS_BIOCATALYSIS database, our tool offers high-accuracy predictions, reflecting the latest in chemical research and data.
Strategy Settings
| Precursor scoring | Relevance Heuristic |
|---|---|
| Min. plausibility | 0.01 |
| Model | Template_relevance |
| Template Set | Pistachio/Bkms_metabolic/Pistachio_ringbreaker/Reaxys/Reaxys_biocatalysis |
| Top-N result to add to graph | 6 |
Feasible Synthetic Routes
試験管内研究製品の免責事項と情報
BenchChemで提示されるすべての記事および製品情報は、情報提供を目的としています。BenchChemで購入可能な製品は、生体外研究のために特別に設計されています。生体外研究は、ラテン語の "in glass" に由来し、生物体の外で行われる実験を指します。これらの製品は医薬品または薬として分類されておらず、FDAから任何の医療状態、病気、または疾患の予防、治療、または治癒のために承認されていません。これらの製品を人間または動物に体内に導入する形態は、法律により厳格に禁止されています。これらのガイドラインに従うことは、研究と実験において法的および倫理的な基準の遵守を確実にするために重要です。


![1,4,6,9-Tetraoxa-5lambda~4~-telluraspiro[4.4]nonane](/img/structure/B89650.png)