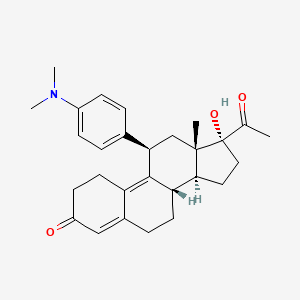
ウリプリスタル
概要
説明
ウリプリスタルは、緊急避妊と子宮筋腫の治療に主に使用される選択的プロゲステロン受容体モジュレーターです。 これは、19-ノルプロゲステロンの誘導体であり、プロゲステロン受容体において拮抗作用と部分的アゴニスト作用の両方を示します .
科学的研究の応用
Ulipristal has a wide range of scientific research applications:
Chemistry: Used as a model compound to study selective progesterone receptor modulators.
Biology: Investigated for its effects on cellular pathways and receptor binding.
Medicine: Primarily used for emergency contraception and the treatment of uterine fibroids.
Industry: Utilized in the pharmaceutical industry for the development of contraceptive pills and fibroid treatments .
作用機序
ウリプリスタルは、プロゲステロン受容体に結合することで効果を発揮し、拮抗作用と部分的アゴニスト作用の両方として作用します。 この結合は、卵胞の破裂を防ぐことで排卵を抑制し、胚の着床を阻止するために子宮内膜に影響を与える可能性もあります . 分子標的は、プロゲステロン受容体と、より少ない程度では、グルココルチコイド受容体です .
生化学分析
Biochemical Properties
Ulipristal plays a significant role in biochemical reactions by interacting with various enzymes, proteins, and other biomolecules. It binds to the progesterone receptor, exhibiting both antagonistic and partial agonist activities. Additionally, ulipristal binds to the glucocorticoid receptor, although it has lower glucocorticoid activity compared to mifepristone . These interactions are crucial for its function as an emergency contraceptive and in the treatment of uterine fibroids.
Cellular Effects
Ulipristal affects various types of cells and cellular processes. In the treatment of uterine fibroids, ulipristal reduces the size of fibroids by inhibiting cell proliferation and inducing apoptosis . It also influences cell signaling pathways, gene expression, and cellular metabolism. For instance, ulipristal has been shown to delay or inhibit ovulation by affecting the endometrium, which may prevent embryo implantation .
Molecular Mechanism
The molecular mechanism of ulipristal involves its binding to the progesterone receptor, where it acts as both an antagonist and partial agonist . This binding prevents progestin from binding to the receptor, thereby inhibiting or delaying ovulation . Ulipristal also alters the normal endometrium, impairing implantation . Additionally, it inhibits the translocation of the glucocorticoid receptor to the nucleus, affecting gene expression .
Temporal Effects in Laboratory Settings
In laboratory settings, the effects of ulipristal change over time. Ulipristal is rapidly absorbed, with a peak plasma concentration occurring approximately one hour after ingestion . Its stability and degradation over time have been studied, showing that it can inhibit follicular rupture and induce early endometrial bleeding in a dose-dependent manner . Long-term effects on cellular function have also been observed, including changes in endometrial maturation and progesterone-dependent markers of implantation .
Dosage Effects in Animal Models
The effects of ulipristal vary with different dosages in animal models. A single post-ovulatory dose of ulipristal has been shown to impair post-fertilization events in mice, reducing the number of conceptuses and early implantation sites . High doses of ulipristal can cause histological and functional alterations in the uterine horns, affecting embryo-uterine interaction .
Metabolic Pathways
Ulipristal is metabolized primarily by the enzyme CYP3A4 and to a lesser extent by CYP1A2 . It is converted into mono-demethylated (active) and di-methylated (inactive) metabolites . These metabolic pathways are crucial for its pharmacokinetics and overall efficacy.
Transport and Distribution
Ulipristal is transported and distributed within cells and tissues, targeting the uterus, cervix, ovaries, and hypothalamus . It binds to plasma proteins with a high affinity, which influences its distribution and localization within the body . The transporters and binding proteins involved in its distribution play a significant role in its therapeutic effects.
Subcellular Localization
The subcellular localization of ulipristal involves its interaction with the glucocorticoid receptor, where it inhibits the receptor’s translocation to the nucleus . This inhibition affects the receptor’s activity and function, leading to changes in gene expression and cellular responses. Ulipristal’s targeting signals and post-translational modifications direct it to specific compartments or organelles, influencing its overall activity.
準備方法
合成経路と反応条件
ウリプリスタルの合成は、19-ノルプロゲステロンから始まり、複数の段階を踏みます反応条件には、通常、付加反応にメチルリチウムまたはメチルグリニャール試薬などの試薬を使用し、その後、加水分解と結晶化の段階で最終生成物を精製します .
工業生産方法
ウリプリスタルの工業生産には、高収率と高純度を確保するために、合成経路の最適化が含まれます。 これには、目的の生成物を得るために、酸性度、温度、反応時間などの加水分解条件を制御することが含まれます。 最終生成物は、エタノールやイソプロパノールなどの溶媒を使用して精製されます .
化学反応の分析
反応の種類
ウリプリスタルは、次のようなさまざまな化学反応を起こします。
酸化: ウリプリスタルは、酸化されてさまざまな代謝産物を形成することができます。
還元: 還元反応は、分子に存在するケトン基を変えることができます。
置換: 置換反応は、ジメチルアミノ基またはアセトキシ基で起こる可能性があります。
一般的な試薬と条件
これらの反応で使用される一般的な試薬には、過マンガン酸カリウムなどの酸化剤と、水素化ホウ素ナトリウムなどの還元剤が含まれます。 反応は、通常、目的の生成物の生成を確実にするために、制御された温度とpH条件で行われます .
生成される主な生成物
これらの反応から生成される主な生成物には、ウリプリスタルの基本構造を保持していますが、特定の官能基に修飾が加えられたさまざまな代謝産物があります .
科学研究への応用
ウリプリスタルは、幅広い科学研究の用途があります。
化学: 選択的プロゲステロン受容体モジュレーターの研究のためのモデル化合物として使用されます。
生物学: 細胞経路と受容体結合への影響について調査されています。
類似化合物との比較
類似化合物
ミフェプリストン: 医学的妊娠中絶と緊急避妊に使用される別のプロゲステロン受容体拮抗薬です。
レボノルゲストレル: 緊急避妊とホルモン避妊薬に使用される合成プロゲステロンです。
ウリプリスタルの独自性
ウリプリスタルは、プロゲステロン受容体において拮抗作用と部分的アゴニスト作用の両方を持つという二重の作用でユニークであり、ミフェプリストンやレボノルゲストレルなどの他の化合物と比較して、独自の有効性と副作用プロファイルを提供します .
特性
IUPAC Name |
(8S,11R,13S,14S,17R)-17-acetyl-11-[4-(dimethylamino)phenyl]-17-hydroxy-13-methyl-1,2,6,7,8,11,12,14,15,16-decahydrocyclopenta[a]phenanthren-3-one | |
|---|---|---|
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
InChI |
InChI=1S/C28H35NO3/c1-17(30)28(32)14-13-25-23-11-7-19-15-21(31)10-12-22(19)26(23)24(16-27(25,28)2)18-5-8-20(9-6-18)29(3)4/h5-6,8-9,15,23-25,32H,7,10-14,16H2,1-4H3/t23-,24+,25-,27-,28-/m0/s1 | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
InChI Key |
HKDLNTKNLJPAIY-WKWWZUSTSA-N | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
Canonical SMILES |
CC(=O)C1(CCC2C1(CC(C3=C4CCC(=O)C=C4CCC23)C5=CC=C(C=C5)N(C)C)C)O | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
Isomeric SMILES |
CC(=O)[C@]1(CC[C@@H]2[C@@]1(C[C@@H](C3=C4CCC(=O)C=C4CC[C@@H]23)C5=CC=C(C=C5)N(C)C)C)O | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
Molecular Formula |
C28H35NO3 | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
DSSTOX Substance ID |
DTXSID501025842 | |
| Record name | Ulipristal | |
| Source | EPA DSSTox | |
| URL | https://comptox.epa.gov/dashboard/DTXSID501025842 | |
| Description | DSSTox provides a high quality public chemistry resource for supporting improved predictive toxicology. | |
Molecular Weight |
433.6 g/mol | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
Mechanism of Action |
The exact mechanism of action of ulipristal has been heavily debated. On one hand, the majority of official prescribing information labels, monographs, and prior research studies for ulipristal indicated as an emergency contraceptive suggest that its primary mechanism of action revolves around inhibiting or delaying ovulation by suppressing surges in LH that result in the postponement of follicular rupture. Conversely, some of the latest investigations pertaining to ulipristal's mechanism of action as an emergency contraceptive propose that it principally elicits its action by preventing embryo implantation, as opposed to preventing ovulation. Although previous investigations have shown that ulipristal essentially has the ability to prevent ovulation equivalent to placebo (ie. null effect or ability) when administered during LH peaks one to two days before ovulation, the agent still demonstrates a stable and consistently high contraceptive effect of approximately >=80% when used at this time. Subsequently, current studies attempt to investigate how ulipristal could elicit emergency contraception via ovulation prevention under circumstances where ovulation had already clearly been observed. Endometrial biopsy samples studied from such circumstances in such investigations subsequently show that the administered ulipristal causes endometrial tissue to become inhospitable and unsuitable for embryo implantation where a variety of genes characteristic of receptive, pro-gestational endometrium are downregulated. Nevertheless, most if not all proposed mechanisms commonly agree that ulipristal ultimately demonstrates its pharmacological effects by binding to human progesterone receptors and prevents natural, endogenous progesterone from occupying such receptors. Regardless, however, considering current and on-going research into ulipristal's ability to prevent embryo implantation, the notion that the medication can elicit post-fertilization effects potentially raises alerts and/or ethical debates over the use of ulipristal owing to potential abortifacient activity, which is considered to be on par or equipotent to that of mifepristone. Attention should be drawn to the fact that some prescribing information, however, such as the US FDA label for ulipristal indicated for emergency contraception, has included new supplementary commentary since 2018 that directly warns about ulipristal not being indicated for termination of existing pregnancies and suggesting that ulipristal use may confer alterations to the endometrium that may affect implantation and contribute to efficacy. In the treatment of fibroids, ulipristal has been shown to exert direct actions on fibroids reducing their size through inhibition of cell proliferation and induction of apoptosis. | |
| Record name | Ulipristal | |
| Source | DrugBank | |
| URL | https://www.drugbank.ca/drugs/DB08867 | |
| Description | The DrugBank database is a unique bioinformatics and cheminformatics resource that combines detailed drug (i.e. chemical, pharmacological and pharmaceutical) data with comprehensive drug target (i.e. sequence, structure, and pathway) information. | |
| Explanation | Creative Common's Attribution-NonCommercial 4.0 International License (http://creativecommons.org/licenses/by-nc/4.0/legalcode) | |
CAS No. |
159811-51-5 | |
| Record name | Ulipristal [USAN:INN:BAN] | |
| Source | ChemIDplus | |
| URL | https://pubchem.ncbi.nlm.nih.gov/substance/?source=chemidplus&sourceid=0159811515 | |
| Description | ChemIDplus is a free, web search system that provides access to the structure and nomenclature authority files used for the identification of chemical substances cited in National Library of Medicine (NLM) databases, including the TOXNET system. | |
| Record name | Ulipristal | |
| Source | DrugBank | |
| URL | https://www.drugbank.ca/drugs/DB08867 | |
| Description | The DrugBank database is a unique bioinformatics and cheminformatics resource that combines detailed drug (i.e. chemical, pharmacological and pharmaceutical) data with comprehensive drug target (i.e. sequence, structure, and pathway) information. | |
| Explanation | Creative Common's Attribution-NonCommercial 4.0 International License (http://creativecommons.org/licenses/by-nc/4.0/legalcode) | |
| Record name | Ulipristal | |
| Source | EPA DSSTox | |
| URL | https://comptox.epa.gov/dashboard/DTXSID501025842 | |
| Description | DSSTox provides a high quality public chemistry resource for supporting improved predictive toxicology. | |
| Record name | (8S,11R,13S,14S,17R)-17-acetyl-11-[4-(dimethylamino)phenyl]-17-hydroxy-13-methyl-1,2,6,7,8,11,12,14,15,16-decahydrocyclopenta[a]phenanthren-3-one | |
| Source | European Chemicals Agency (ECHA) | |
| URL | https://echa.europa.eu/information-on-chemicals | |
| Description | The European Chemicals Agency (ECHA) is an agency of the European Union which is the driving force among regulatory authorities in implementing the EU's groundbreaking chemicals legislation for the benefit of human health and the environment as well as for innovation and competitiveness. | |
| Explanation | Use of the information, documents and data from the ECHA website is subject to the terms and conditions of this Legal Notice, and subject to other binding limitations provided for under applicable law, the information, documents and data made available on the ECHA website may be reproduced, distributed and/or used, totally or in part, for non-commercial purposes provided that ECHA is acknowledged as the source: "Source: European Chemicals Agency, http://echa.europa.eu/". Such acknowledgement must be included in each copy of the material. ECHA permits and encourages organisations and individuals to create links to the ECHA website under the following cumulative conditions: Links can only be made to webpages that provide a link to the Legal Notice page. | |
| Record name | ULIPRISTAL | |
| Source | FDA Global Substance Registration System (GSRS) | |
| URL | https://gsrs.ncats.nih.gov/ginas/app/beta/substances/6J5J15Q2X8 | |
| Description | The FDA Global Substance Registration System (GSRS) enables the efficient and accurate exchange of information on what substances are in regulated products. Instead of relying on names, which vary across regulatory domains, countries, and regions, the GSRS knowledge base makes it possible for substances to be defined by standardized, scientific descriptions. | |
| Explanation | Unless otherwise noted, the contents of the FDA website (www.fda.gov), both text and graphics, are not copyrighted. They are in the public domain and may be republished, reprinted and otherwise used freely by anyone without the need to obtain permission from FDA. Credit to the U.S. Food and Drug Administration as the source is appreciated but not required. | |
Retrosynthesis Analysis
AI-Powered Synthesis Planning: Our tool employs the Template_relevance Pistachio, Template_relevance Bkms_metabolic, Template_relevance Pistachio_ringbreaker, Template_relevance Reaxys, Template_relevance Reaxys_biocatalysis model, leveraging a vast database of chemical reactions to predict feasible synthetic routes.
One-Step Synthesis Focus: Specifically designed for one-step synthesis, it provides concise and direct routes for your target compounds, streamlining the synthesis process.
Accurate Predictions: Utilizing the extensive PISTACHIO, BKMS_METABOLIC, PISTACHIO_RINGBREAKER, REAXYS, REAXYS_BIOCATALYSIS database, our tool offers high-accuracy predictions, reflecting the latest in chemical research and data.
Strategy Settings
| Precursor scoring | Relevance Heuristic |
|---|---|
| Min. plausibility | 0.01 |
| Model | Template_relevance |
| Template Set | Pistachio/Bkms_metabolic/Pistachio_ringbreaker/Reaxys/Reaxys_biocatalysis |
| Top-N result to add to graph | 6 |
Feasible Synthetic Routes
試験管内研究製品の免責事項と情報
BenchChemで提示されるすべての記事および製品情報は、情報提供を目的としています。BenchChemで購入可能な製品は、生体外研究のために特別に設計されています。生体外研究は、ラテン語の "in glass" に由来し、生物体の外で行われる実験を指します。これらの製品は医薬品または薬として分類されておらず、FDAから任何の医療状態、病気、または疾患の予防、治療、または治癒のために承認されていません。これらの製品を人間または動物に体内に導入する形態は、法律により厳格に禁止されています。これらのガイドラインに従うことは、研究と実験において法的および倫理的な基準の遵守を確実にするために重要です。


![5-(2,2-Diphenylacetyl)-4-[(4-methoxy-3-methylphenyl)methyl]-1,4,6,7-tetrahydroimidazo[4,5-c]pyridine-6-carboxylic acid](/img/structure/B1683315.png)
![(2S)-2-[[2-[[2-[[(2S)-2-[[(2S)-4-amino-2-[[(2R)-3-[2,3-di(hexadecanoyloxy)propylsulfanyl]-2-(hexadecanoylamino)propanoyl]amino]-4-oxobutanoyl]amino]-3-hydroxypropanoyl]amino]acetyl]amino]acetyl]amino]-3-hydroxypropanoic acid](/img/structure/B1683328.png)
![12-[4-(4-pyrimidin-2-ylpiperazin-1-yl)butyl]-12-azapentacyclo[7.5.0.02,7.03,5.04,8]tetradecane-11,13-dione](/img/structure/B1683330.png)