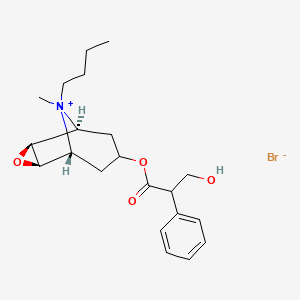
卡麦角林
描述
卡麦角林是一种多巴胺能药物,主要用于治疗与高泌乳素水平相关的疾病,例如泌乳素瘤和高泌乳素血症。 它也用于治疗帕金森病和其他疾病。 卡麦角林是一种麦角衍生物,作为一种强效的多巴胺 D2 受体激动剂 .
科学研究应用
卡麦角林具有广泛的科学研究应用:
医学: 它用于治疗高泌乳素血症性疾病,帕金森病和分泌泌乳素的垂体腺瘤。
作用机制
卡麦角林通过刺激多巴胺 D2 受体发挥作用,多巴胺 D2 受体是与 Gi 蛋白相关的 G 蛋白偶联受体。 在乳腺促素细胞中,刺激多巴胺 D2 受体抑制腺苷酸环化酶,降低细胞内环腺苷酸(cAMP)浓度,并阻断肌醇三磷酸(IP3)依赖性钙从细胞内储存库的释放。 这导致泌乳素分泌的抑制 .
生化分析
Biochemical Properties
Cabergoline interacts with dopamine D2 receptors, exhibiting potent agonist activity . It is extensively metabolized by the liver, predominantly via hydrolysis of the acylurea bond of the urea moiety . Cytochrome P450-mediated metabolism is minimal .
Cellular Effects
Cabergoline has been shown to influence various types of cells and cellular processes. It is used to reduce the production of a hormone called prolactin by the pituitary gland . In human endometrial stromal cells, Cabergoline treatment more than doubled decidual biomarker expression . It also induced characteristic decidual morphology changes and blocked detrimental effects of IL-1β on decidual cytology .
Molecular Mechanism
Cabergoline exerts its effects at the molecular level primarily through its action as a dopamine D2 receptor agonist . The stimulation of dopamine D2 receptors leads to improvements in coordinated muscle activity in those with movement disorders .
Temporal Effects in Laboratory Settings
Cabergoline is a long-acting medication, usually taken once or twice a week . The elimination half-life of cabergoline estimated from urinary data of healthy subjects ranges between 63 and 109 hours .
Dosage Effects in Animal Models
The effects of Cabergoline can vary with different dosages in animal models
Metabolic Pathways
Cabergoline is involved in metabolic pathways predominantly via hydrolysis of the acylurea bond of the urea moiety . The major metabolites identified thus far do not contribute to the therapeutic effect of Cabergoline .
Subcellular Localization
In terms of subcellular localization, one study found strong immunohistochemistry localization of dopamine D2 receptors (which Cabergoline interacts with) in the endometrial stromal compartment with a subcellular distribution distinct from that of ERα .
准备方法
卡麦角林通过一系列化学反应从麦角碱-8β-羧酸酯合成。 该过程包括将麦角碱-8β-羧酸 C1-4 烷基酯的仲胺和吲哚氮官能团保护为氨基甲酸酯衍生物。 然后,将保护的化合物用 3-(二甲氨基)丙胺酰化,与异氰酸乙酯反应,并裂解保护基。 最后,将脱保护的仲胺与亲电烯丙醇衍生物反应,得到卡麦角林 .
化学反应分析
卡麦角林会发生各种化学反应,包括水解和氧化。 它对水解高度敏感,尤其是在尿素部分和酰胺基团。 卡麦角林中的烯烃键易于氧化。 这些反应中常用的试剂包括用于水解的水和用于氧化的氧化剂。 通过红外光谱和质谱分析鉴定出这些反应形成的主要产物为降解产物.
相似化合物的比较
卡麦角林经常与其他多巴胺激动剂进行比较,例如溴隐亭和麦角新碱。 虽然这三种化合物都用于治疗高泌乳素血症和帕金森病,但卡麦角林具有更长的半衰期和更高的多巴胺 D2 受体亲和力,使其更有效,并且更易于患者耐受 . 溴隐亭和麦角新碱具有更短的半衰期,可能需要更频繁的给药 .
类似化合物
- 溴隐亭
- 麦角新碱
- 喹那戈莱德
卡麦角林的独特特性,如其长的半衰期和高受体亲和力,使其成为许多患者和医疗保健提供者的首选药物。
属性
IUPAC Name |
(6aR,9R,10aR)-N-[3-(dimethylamino)propyl]-N-(ethylcarbamoyl)-7-prop-2-enyl-6,6a,8,9,10,10a-hexahydro-4H-indolo[4,3-fg]quinoline-9-carboxamide | |
|---|---|---|
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
InChI |
InChI=1S/C26H37N5O2/c1-5-11-30-17-19(25(32)31(26(33)27-6-2)13-8-12-29(3)4)14-21-20-9-7-10-22-24(20)18(16-28-22)15-23(21)30/h5,7,9-10,16,19,21,23,28H,1,6,8,11-15,17H2,2-4H3,(H,27,33)/t19-,21-,23-/m1/s1 | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
InChI Key |
KORNTPPJEAJQIU-KJXAQDMKSA-N | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
Canonical SMILES |
CCNC(=O)N(CCCN(C)C)C(=O)C1CC2C(CC3=CNC4=CC=CC2=C34)N(C1)CC=C | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
Isomeric SMILES |
CCNC(=O)N(CCCN(C)C)C(=O)[C@@H]1C[C@H]2[C@@H](CC3=CNC4=CC=CC2=C34)N(C1)CC=C | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
Molecular Formula |
C26H37N5O2 | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
Related CAS |
85329-89-1 (diphosphate) | |
| Record name | Cabergoline [USAN:USP:INN:BAN] | |
| Source | ChemIDplus | |
| URL | https://pubchem.ncbi.nlm.nih.gov/substance/?source=chemidplus&sourceid=0081409907 | |
| Description | ChemIDplus is a free, web search system that provides access to the structure and nomenclature authority files used for the identification of chemical substances cited in National Library of Medicine (NLM) databases, including the TOXNET system. | |
DSSTOX Substance ID |
DTXSID6022719 | |
| Record name | Cabergoline | |
| Source | EPA DSSTox | |
| URL | https://comptox.epa.gov/dashboard/DTXSID6022719 | |
| Description | DSSTox provides a high quality public chemistry resource for supporting improved predictive toxicology. | |
Molecular Weight |
451.6 g/mol | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
Physical Description |
Solid | |
| Record name | Cabergoline | |
| Source | Human Metabolome Database (HMDB) | |
| URL | http://www.hmdb.ca/metabolites/HMDB0014393 | |
| Description | The Human Metabolome Database (HMDB) is a freely available electronic database containing detailed information about small molecule metabolites found in the human body. | |
| Explanation | HMDB is offered to the public as a freely available resource. Use and re-distribution of the data, in whole or in part, for commercial purposes requires explicit permission of the authors and explicit acknowledgment of the source material (HMDB) and the original publication (see the HMDB citing page). We ask that users who download significant portions of the database cite the HMDB paper in any resulting publications. | |
Solubility |
Insoluble, 6.40e-02 g/L | |
| Record name | Cabergoline | |
| Source | DrugBank | |
| URL | https://www.drugbank.ca/drugs/DB00248 | |
| Description | The DrugBank database is a unique bioinformatics and cheminformatics resource that combines detailed drug (i.e. chemical, pharmacological and pharmaceutical) data with comprehensive drug target (i.e. sequence, structure, and pathway) information. | |
| Explanation | Creative Common's Attribution-NonCommercial 4.0 International License (http://creativecommons.org/licenses/by-nc/4.0/legalcode) | |
| Record name | Cabergoline | |
| Source | Human Metabolome Database (HMDB) | |
| URL | http://www.hmdb.ca/metabolites/HMDB0014393 | |
| Description | The Human Metabolome Database (HMDB) is a freely available electronic database containing detailed information about small molecule metabolites found in the human body. | |
| Explanation | HMDB is offered to the public as a freely available resource. Use and re-distribution of the data, in whole or in part, for commercial purposes requires explicit permission of the authors and explicit acknowledgment of the source material (HMDB) and the original publication (see the HMDB citing page). We ask that users who download significant portions of the database cite the HMDB paper in any resulting publications. | |
Mechanism of Action |
The dopamine D2 receptor is a 7-transmembrane G-protein coupled receptor associated with Gi proteins. In lactotrophs, stimulation of dopamine D2 causes inhibition of adenylyl cyclase, which decreases intracellular cAMP concentrations and blocks IP3-dependent release of Ca2+ from intracellular stores. Decreases in intracellular calcium levels may also be brought about via inhibition of calcium influx through voltage-gated calcium channels, rather than via inhibition of adenylyl cyclase. Additionally, receptor activation blocks phosphorylation of p42/p44 MAPK and decreases MAPK/ERK kinase phosphorylation. Inhibition of MAPK appears to be mediated by c-Raf and B-Raf-dependent inhibition of MAPK/ERK kinase. Dopamine-stimulated growth hormone release from the pituitary gland is mediated by a decrease in intracellular calcium influx through voltage-gated calcium channels rather than via adenylyl cyclase inhibition. Stimulation of dopamine D2 receptors in the nigrostriatal pathway leads to improvements in coordinated muscle activity in those with movement disorders. Cabergoline is a long-acting dopamine receptor agonist with a high affinity for D2 receptors. Receptor-binding studies indicate that cabergoline has low affinity for dopamine D1, α1,- and α2- adrenergic, and 5-HT1- and 5-HT2-serotonin receptors. | |
| Record name | Cabergoline | |
| Source | DrugBank | |
| URL | https://www.drugbank.ca/drugs/DB00248 | |
| Description | The DrugBank database is a unique bioinformatics and cheminformatics resource that combines detailed drug (i.e. chemical, pharmacological and pharmaceutical) data with comprehensive drug target (i.e. sequence, structure, and pathway) information. | |
| Explanation | Creative Common's Attribution-NonCommercial 4.0 International License (http://creativecommons.org/licenses/by-nc/4.0/legalcode) | |
CAS No. |
81409-90-7 | |
| Record name | Cabergoline | |
| Source | CAS Common Chemistry | |
| URL | https://commonchemistry.cas.org/detail?cas_rn=81409-90-7 | |
| Description | CAS Common Chemistry is an open community resource for accessing chemical information. Nearly 500,000 chemical substances from CAS REGISTRY cover areas of community interest, including common and frequently regulated chemicals, and those relevant to high school and undergraduate chemistry classes. This chemical information, curated by our expert scientists, is provided in alignment with our mission as a division of the American Chemical Society. | |
| Explanation | The data from CAS Common Chemistry is provided under a CC-BY-NC 4.0 license, unless otherwise stated. | |
| Record name | Cabergoline [USAN:USP:INN:BAN] | |
| Source | ChemIDplus | |
| URL | https://pubchem.ncbi.nlm.nih.gov/substance/?source=chemidplus&sourceid=0081409907 | |
| Description | ChemIDplus is a free, web search system that provides access to the structure and nomenclature authority files used for the identification of chemical substances cited in National Library of Medicine (NLM) databases, including the TOXNET system. | |
| Record name | Cabergoline | |
| Source | DrugBank | |
| URL | https://www.drugbank.ca/drugs/DB00248 | |
| Description | The DrugBank database is a unique bioinformatics and cheminformatics resource that combines detailed drug (i.e. chemical, pharmacological and pharmaceutical) data with comprehensive drug target (i.e. sequence, structure, and pathway) information. | |
| Explanation | Creative Common's Attribution-NonCommercial 4.0 International License (http://creativecommons.org/licenses/by-nc/4.0/legalcode) | |
| Record name | Cabergoline | |
| Source | EPA DSSTox | |
| URL | https://comptox.epa.gov/dashboard/DTXSID6022719 | |
| Description | DSSTox provides a high quality public chemistry resource for supporting improved predictive toxicology. | |
| Record name | (6aR,9R,10aR)-N-[3-(dimethylamino)propyl]-N-(ethylcarbamoyl)-7-prop-2-enyl-6,6a,8,9,10,10a-hexahydro-4H-indolo[4,3-fg]quinoline-9-carboxamide | |
| Source | European Chemicals Agency (ECHA) | |
| URL | https://echa.europa.eu/information-on-chemicals | |
| Description | The European Chemicals Agency (ECHA) is an agency of the European Union which is the driving force among regulatory authorities in implementing the EU's groundbreaking chemicals legislation for the benefit of human health and the environment as well as for innovation and competitiveness. | |
| Explanation | Use of the information, documents and data from the ECHA website is subject to the terms and conditions of this Legal Notice, and subject to other binding limitations provided for under applicable law, the information, documents and data made available on the ECHA website may be reproduced, distributed and/or used, totally or in part, for non-commercial purposes provided that ECHA is acknowledged as the source: "Source: European Chemicals Agency, http://echa.europa.eu/". Such acknowledgement must be included in each copy of the material. ECHA permits and encourages organisations and individuals to create links to the ECHA website under the following cumulative conditions: Links can only be made to webpages that provide a link to the Legal Notice page. | |
| Record name | CABERGOLINE | |
| Source | FDA Global Substance Registration System (GSRS) | |
| URL | https://gsrs.ncats.nih.gov/ginas/app/beta/substances/LL60K9J05T | |
| Description | The FDA Global Substance Registration System (GSRS) enables the efficient and accurate exchange of information on what substances are in regulated products. Instead of relying on names, which vary across regulatory domains, countries, and regions, the GSRS knowledge base makes it possible for substances to be defined by standardized, scientific descriptions. | |
| Explanation | Unless otherwise noted, the contents of the FDA website (www.fda.gov), both text and graphics, are not copyrighted. They are in the public domain and may be republished, reprinted and otherwise used freely by anyone without the need to obtain permission from FDA. Credit to the U.S. Food and Drug Administration as the source is appreciated but not required. | |
| Record name | Cabergoline | |
| Source | Human Metabolome Database (HMDB) | |
| URL | http://www.hmdb.ca/metabolites/HMDB0014393 | |
| Description | The Human Metabolome Database (HMDB) is a freely available electronic database containing detailed information about small molecule metabolites found in the human body. | |
| Explanation | HMDB is offered to the public as a freely available resource. Use and re-distribution of the data, in whole or in part, for commercial purposes requires explicit permission of the authors and explicit acknowledgment of the source material (HMDB) and the original publication (see the HMDB citing page). We ask that users who download significant portions of the database cite the HMDB paper in any resulting publications. | |
Melting Point |
102-104 °C, 102 - 104 °C | |
| Record name | Cabergoline | |
| Source | DrugBank | |
| URL | https://www.drugbank.ca/drugs/DB00248 | |
| Description | The DrugBank database is a unique bioinformatics and cheminformatics resource that combines detailed drug (i.e. chemical, pharmacological and pharmaceutical) data with comprehensive drug target (i.e. sequence, structure, and pathway) information. | |
| Explanation | Creative Common's Attribution-NonCommercial 4.0 International License (http://creativecommons.org/licenses/by-nc/4.0/legalcode) | |
| Record name | Cabergoline | |
| Source | Human Metabolome Database (HMDB) | |
| URL | http://www.hmdb.ca/metabolites/HMDB0014393 | |
| Description | The Human Metabolome Database (HMDB) is a freely available electronic database containing detailed information about small molecule metabolites found in the human body. | |
| Explanation | HMDB is offered to the public as a freely available resource. Use and re-distribution of the data, in whole or in part, for commercial purposes requires explicit permission of the authors and explicit acknowledgment of the source material (HMDB) and the original publication (see the HMDB citing page). We ask that users who download significant portions of the database cite the HMDB paper in any resulting publications. | |
Retrosynthesis Analysis
AI-Powered Synthesis Planning: Our tool employs the Template_relevance Pistachio, Template_relevance Bkms_metabolic, Template_relevance Pistachio_ringbreaker, Template_relevance Reaxys, Template_relevance Reaxys_biocatalysis model, leveraging a vast database of chemical reactions to predict feasible synthetic routes.
One-Step Synthesis Focus: Specifically designed for one-step synthesis, it provides concise and direct routes for your target compounds, streamlining the synthesis process.
Accurate Predictions: Utilizing the extensive PISTACHIO, BKMS_METABOLIC, PISTACHIO_RINGBREAKER, REAXYS, REAXYS_BIOCATALYSIS database, our tool offers high-accuracy predictions, reflecting the latest in chemical research and data.
Strategy Settings
| Precursor scoring | Relevance Heuristic |
|---|---|
| Min. plausibility | 0.01 |
| Model | Template_relevance |
| Template Set | Pistachio/Bkms_metabolic/Pistachio_ringbreaker/Reaxys/Reaxys_biocatalysis |
| Top-N result to add to graph | 6 |
Feasible Synthetic Routes
Q1: What is the primary mechanism of action of Cabergoline?
A: Cabergoline exerts its therapeutic effect primarily by acting as a dopamine receptor agonist, exhibiting a high affinity for the dopamine D2 receptor subtype. [, , ] This interaction mimics the action of dopamine, a neurotransmitter, leading to a cascade of downstream effects.
Q2: How does Cabergoline's interaction with dopamine D2 receptors affect prolactin secretion?
A: Prolactin secretion from the pituitary gland is under tonic inhibitory control by dopamine. By binding to D2 receptors on lactotroph cells in the pituitary gland, Cabergoline inhibits the synthesis and release of prolactin. [, , , ]
Q3: Beyond prolactin, what other hormonal or physiological processes are influenced by Cabergoline?
A: Cabergoline's action on dopamine receptors extends beyond prolactin regulation. It can also affect growth hormone secretion, potentially finding application in managing acromegaly. [, , ] Additionally, its influence on the dopaminergic system suggests potential applications in treating conditions like Parkinson's disease. [, , , ]
Q4: What is the molecular formula and weight of Cabergoline?
A: Cabergoline is represented by the molecular formula C26H37N5O2 and has a molecular weight of 451.6 g/mol. []
Q5: Is there any available spectroscopic data for Cabergoline?
A: While the provided research doesn't delve into specific spectroscopic data, techniques like nuclear magnetic resonance (NMR) and mass spectrometry (MS) are commonly employed for the structural characterization of compounds like Cabergoline. []
Q6: Is there information available regarding the material compatibility and stability of Cabergoline under various conditions?
A6: The provided research focuses primarily on Cabergoline's pharmacological properties and applications in biological systems. Specific details regarding its material compatibility and stability in non-biological contexts are not discussed.
Q7: Are there any known factors that influence the pharmacokinetics of Cabergoline?
A: Concomitant administration of drugs like clarithromycin, a potent inhibitor of CYP3A4 (a key enzyme involved in drug metabolism), can significantly increase the blood concentration of Cabergoline. []
Q8: What are the primary clinical applications of Cabergoline?
A: Cabergoline is primarily prescribed for the treatment of hyperprolactinemia, often caused by prolactinomas. [, , , , ] Its efficacy in normalizing prolactin levels, restoring ovulation and fertility in women, and reducing tumor size has been well-documented. [, , , , ]
Q9: Can Cabergoline be used during pregnancy?
A: While the use of Cabergoline during pregnancy has been reported, and available data suggests it may be relatively safe, bromocriptine, another dopamine agonist, has a larger safety database and a more established safety record for use during pregnancy. []
Q10: Is Cabergoline effective in treating conditions other than hyperprolactinemia?
A: Emerging research suggests potential applications for Cabergoline in managing acromegaly, either as monotherapy or in combination with other drugs like pegvisomant. [, , ] Additionally, its use in treating Parkinson's disease and restless legs syndrome has been explored. [, , , ]
Q11: Are there alternative treatment options for individuals with Cabergoline-resistant prolactinomas?
A: In cases of Cabergoline resistance, alternative dopamine agonists like bromocriptine might be considered, although they often have a less favorable side-effect profile. [] Other options include surgery or radiotherapy. [, ]
Q12: Are there any specific drug delivery or targeting strategies being explored for Cabergoline?
A12: The provided research primarily focuses on Cabergoline's systemic effects. Targeted drug delivery strategies are not extensively discussed within this context.
Q13: What analytical methods are commonly used to characterize and quantify Cabergoline?
A: Techniques like high-performance liquid chromatography (HPLC) coupled with mass spectrometry (MS) are commonly employed to analyze Cabergoline and its metabolites. []
Q14: Is there information available on the environmental impact and degradation of Cabergoline?
A14: The provided research primarily focuses on the pharmacological aspects of Cabergoline in treating human conditions. Environmental impact and degradation are not discussed within this scope.
Q15: Are there any cross-disciplinary applications or research synergies related to Cabergoline?
A: Research on Cabergoline spans various disciplines, including endocrinology, neurology, and pharmacology. Its mechanisms of action and effects on hormone regulation have implications for understanding pituitary gland function and dopamine signaling pathways. [, , , , , , , , , , , , , , ]
体外研究产品的免责声明和信息
请注意,BenchChem 上展示的所有文章和产品信息仅供信息参考。 BenchChem 上可购买的产品专为体外研究设计,这些研究在生物体外进行。体外研究,源自拉丁语 "in glass",涉及在受控实验室环境中使用细胞或组织进行的实验。重要的是要注意,这些产品没有被归类为药物或药品,他们没有得到 FDA 的批准,用于预防、治疗或治愈任何医疗状况、疾病或疾病。我们必须强调,将这些产品以任何形式引入人类或动物的身体都是法律严格禁止的。遵守这些指南对确保研究和实验的法律和道德标准的符合性至关重要。